Not content with using hybrid artificial photosynthesis to turn CO2 emissions into plastics and biofuel, researchers at the Lawrence Berkeley National Laboratory (Berkeley Lab) now claim to have produced an enhanced system that uses water and solar energy to generate hydrogen, which is in turn used to produce methane, the main element of natural gas, from carbon dioxide. Generating such gases from a renewable resource may one day help bolster, or even replace, fossil fuel resources extracted from dwindling sub-surface deposits.
Simply put, the process of photosynthesis turns light energyinto chemical energy. In plants and certain types of algae, energy fromincoming sunlight is used as the power source to synthesize simplecarbohydrates from carbon dioxide and water. In the original BerkeleyLab hybrid system, a membrane arrangement of nanowires created from silicon andtitanium oxide harvested solar energy and transported electrons to microbeswhere they used that energy to transform carbon dioxide into a range ofchemical compounds.
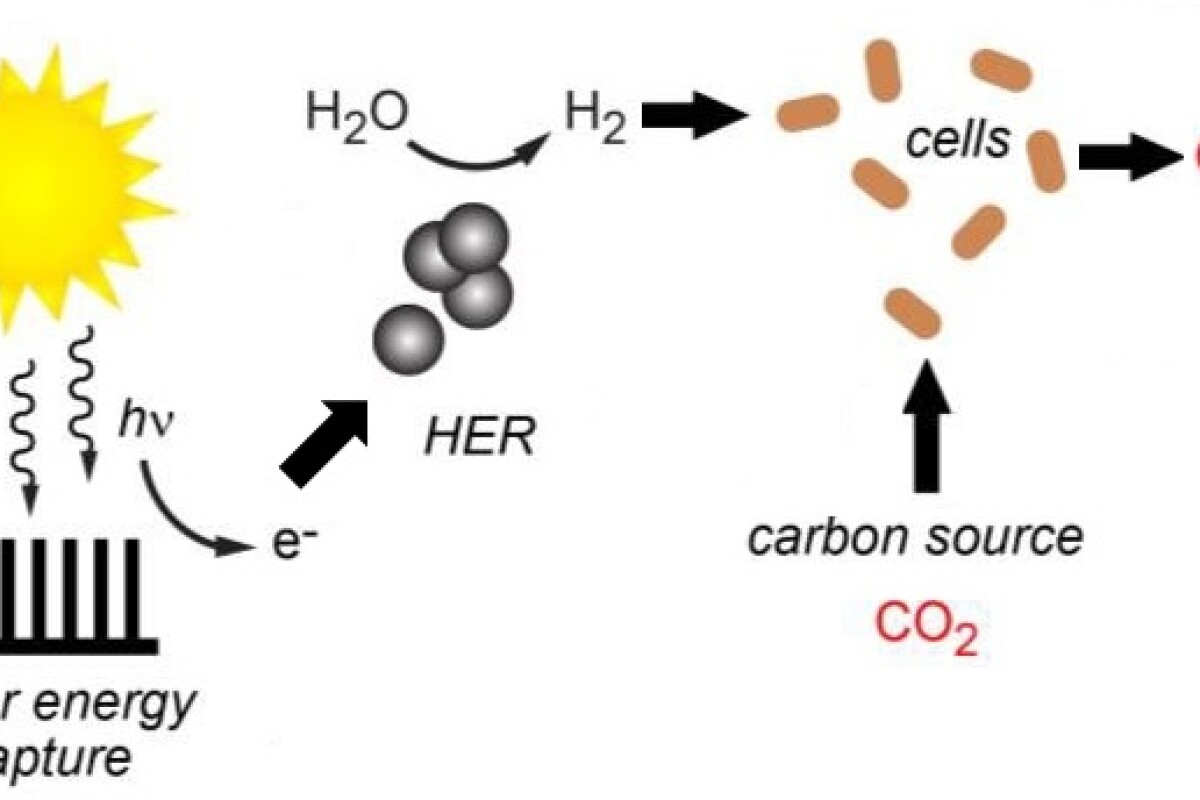
In the latest iteration of the artificial photosynthesis system, solar energy was captured via a similar membrane (but this timeconsisting of indium phosphide photocathodes and titanium dioxide photoanodes),which was employed to supply power for the splitting of water moleculesinto oxygen and hydrogen. The hydrogen was then conveyed to a collection ofmicrobes that used it to convert carbon dioxide into methane. Hence thehybrid system collected light energy and produced both hydrogen and methane.
"This study representsanother key breakthrough in solar-to-chemical energy conversion efficiency andartificial photosynthesis," said Professor Peidong Yang, a chemist withBerkeley Lab’s Materials Sciences Division. "By generating renewablehydrogen and feeding it to microbes for the production of methane, we can nowexpect an electrical-to-chemical efficiency of better than 50 percent and asolar-to-chemical energy conversion efficiency of 10-percent if our system iscoupled with state-of-art solar panel and electrolyzer."

Though the fundamentalconcept in the two artificial photosynthesis experiments is largely similar, inthe first tranche of work the researchers used an anaerobic bacterium, Sporomusa ovata, to transform carbondioxide when fed with electrons. In the latest iteration, the scientistspopulated the membrane with Methanosarcinabarkeri, which is an anaerobic archaeon (essentially a single-celledmicroorganism that has no cell nucleus or other membrane-bound organelles) thattransforms carbon dioxide using hydrogen itself.
In this way, water is turnedinto hydrogen by a hydrogen evolution reaction (HER), where the HER iscatalyzed by the addition of nickel sulfide nanoparticles thatoperate effectively under biologically compatible conditions.
"Using hydrogen as theenergy carrier rather than electrons makes for a much more efficient process asmolecular hydrogen, through its chemical bonds, has a much higher density forstoring and transporting energy," said Associate Professor of chemistry atBerkeley Lab, and member of the research team, Michelle Chang.
"While we were inspiredby the process of natural photosynthesis and continue to learn from it, byadding nanotechnology to help improve the efficiency of natural systems we areshowing that sometimes we can do even better than nature," added ProfessorYang.
Whilst this research is amulti-pronged approach to producing a range of gases and chemicals, it is alsoa method that brings living organisms into the mix. As such, even though purely electricalmethods of solar hydrogen production are increasing in efficiency, and it is possible to use solar energy combined with cheap and abundant mineral elements to create hydrogen, theidea of generating a range of useful, energy-rich gases using justsunlight, water, CO2 and naturally-occurring microbes in a process scaled-up tocommercial sizes holds a great deal of appeal in creating a trulyenvironmentally-friendly and self-sufficient energy production system.
"Weselected methane as an initial target owing to the ease of product separation,the potential for integration into existing infrastructures for the deliveryand use of natural gas, and the fact that direct conversion of carbon dioxideto methane with synthetic catalysts has proven to be a formidable challenge,"said Chris Chang, another professor of chemistry at Berkeley Lab and a member of the research team. "Since we still get the majority of our methane from naturalgas, a fossil fuel, often from fracking, the ability to generate methane from arenewable hydrogen source is another important advance."
The results of this latest research were recently published in the journal Proceedings of the National Academy of Sciences (PNAS).
Source: Berkeley Lab