For many people the problem of Primary Axillary Hyperhidrosis or excessive underarm sweating is a huge issue affecting their quality of life. This medical condition occurs when the sweat glands are overactive in creating more sweat than is needed to cool the body – sometimes four or five times more sweat than normal. Miramar Labs has developed a new solution to this problem in the form of the miraDry system – a treatment that uses microwave energy to deactivate the sweat glands in the underarm.
Sweating is the mechanism that your body uses to regulate its body temperature. The human body has over 2 million sweat glands all over the body to help you do this. Even though you do not feel it, they are all contributing to help maintain your body temperature.
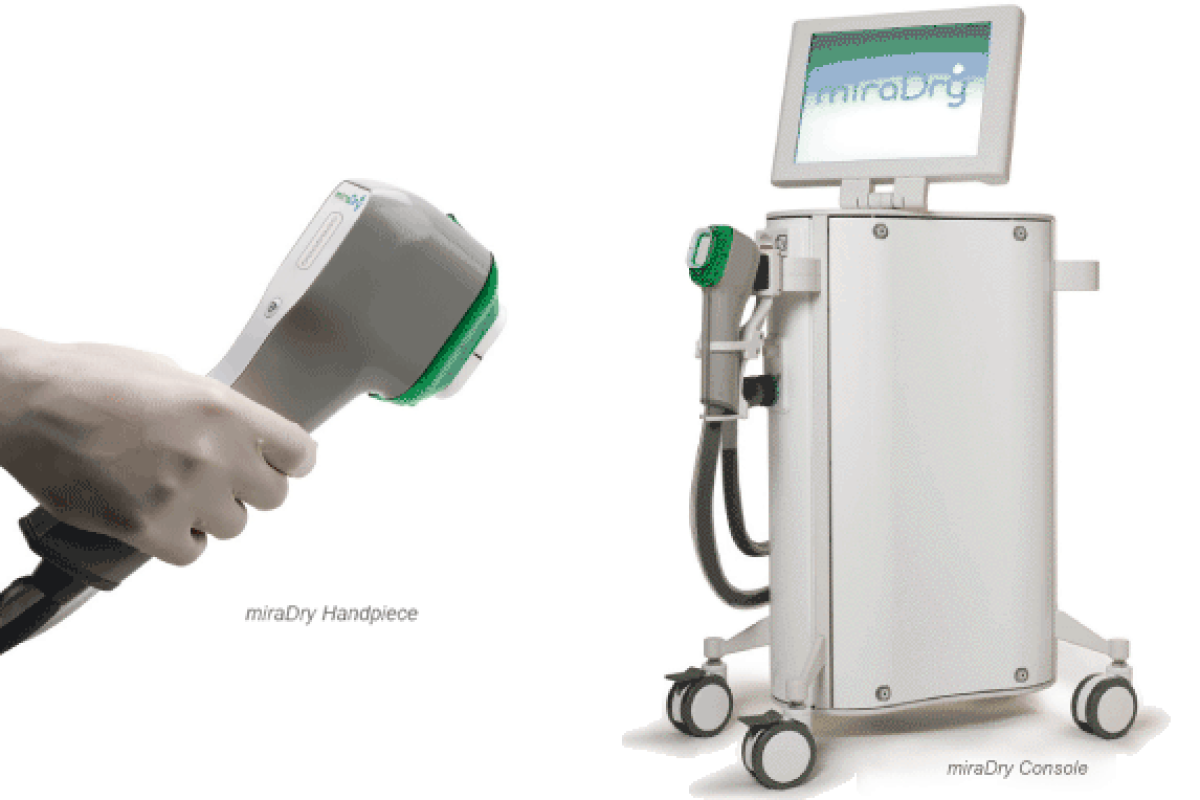
The miraDry system consists of a console, handpiece and a disposable tip. Billed as secure and long-lasting, the non-invasive treatment delivers electromagnetic energy to target and deactivate the sweat glands in the underarm. Firstly, the underarm is numbed using local anesthesia. The miraDry handpiece then delivers microwave energy to the area causing thermal necrosis of the sweat glands. The procedure takes 40 minutes and performed in one or two visits.
According to Miramar Labs, switching off the 2% of sweat glands that are located in the underarms will not affect the body's temperature regulating function. Patients can expect to experience some soreness for a few days with mild to moderate swelling common, lasting a week or two, but the company says that a positive effect will be noticed immediately following the procedure.

Current alternatives for the treatment of Primary Axillary Hyperhidrosis include prescription antiperspirants or for more severe cases, multiple injections of Botulinum toxin type A in the underarms every six months. With both these solutions, symptoms will return. Surgery – with its inherent risks – is the other option for severe cases involving the removal of the sweat glands.
Miramar Labs recently announced that it has received FDA 510(k) clearance for the miraDry System.