Oil and water don't mix, but they can make a messy froth that makes it difficult to separate the two. In an effort to keep the two separated, engineers at MIT are literally shining a light on the problem. Using special organic dyes, the researchers have come up with a system that controls how water moves on a surface, which may one day help clean up oil drilling and also holds the promise of creating microfluidic diagnostic devices that can be redesigned at the push of a button.
Drilling for oil is a dirty business. One particularly unpleasant problem is when oil and water try to mix. Some types of drilling use an injection of brine into the well and what comes up is often briny water mixed with oil droplets in a fine, frothy suspension. According to MIT, there are ways of separating this brown-black foam, including the use of electrostatic devices, but they tend to be energy hogs and aren't effective if the water is too saline.
The MIT team's approach involved looking at a different type of oil – suntan oil. Most sunscreens use titanium dioxide as their active ingredient to protect against the sun, and it has a bit of a Jekyll and Hyde nature. When in the dark or exposed to visible light, titanium oxide repels water, but when it's exposed to ultraviolet light, it attracts water and becomes wettable.

This phenomenon, called photoresponsiveness, formed the basis for the new MIT system. The researchers surmised that if they could get a surface to become wettable on command by shining a light on it, they could directly separate the oil from the water by using light to drive the water droplets together until they formed a single film, which would split off from the oil like salad dressing left to sit in the bottle.
The only problem was that most photoresponsive materials react to ultraviolet light, not visible. But the team found that by binding titanium oxide to a polymer film on a layer of glass, then dipping it in an organic dye, it would become highly responsive to visible light – much more so than to UV, which makes up only five percent of the spectrum.
By putting the material out in the sun, it acted as a very good oil/water separator to break up the foam. In tests, almost all the salt water spread out on the surface as it separated from the oil.
"We were inspired by the work in photovoltaics, where dye sensitization was used to improve the efficiency of absorption of solar radiation," says MIT associate professor of mechanical engineering Kripa Varanasi. "The coupling of the dye to [titanium oxide] particles allows for the generation of charge carriers upon light illumination. This creates an electric potential difference to be established between the surface and the liquid upon illumination, and leads to a change in the wetting properties."
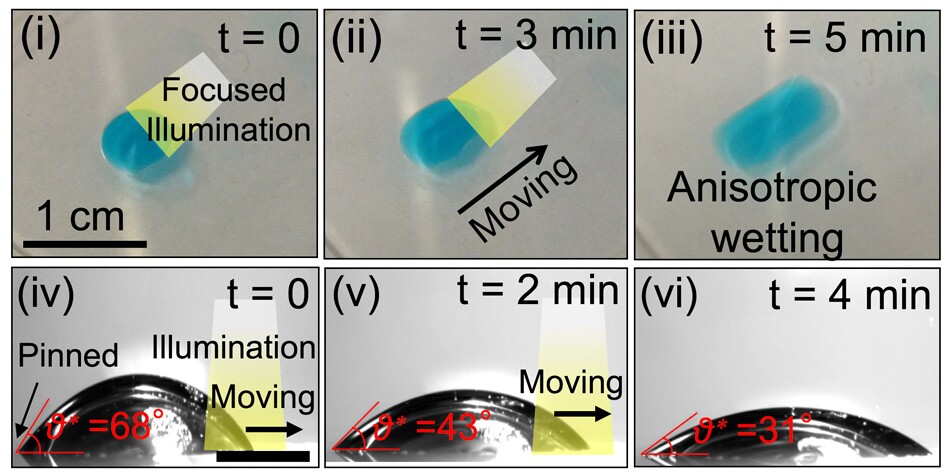
Experiments on the new technology also found that by shining a beam of light on the photoresponsive surface, selected areas became momentarily wettable. This allowed the researchers to move droplets along with great precision.

According to MIT, this would allow engineers to design a microfluidic lab-on-a-chip that wouldn't need any built-in structure. Instead, patterns of light could create the needed boundaries to move droplets around for analysis. In addition, the ability to turn wettability on and off would make the surfaces of such devices self-cleaning as they suddenly repelled water and carried away contaminants. Furthermore, using different dyes would make the surfaces very customizable and inexpensive.
"By systematically studying the relationship between the energy levels of the dye and the wettability of the contacting liquid, we have come up with a framework for the design of these light-guided liquid manipulation systems," says Varanasi. "By choosing the right kind of dye, we can create a significant change in droplet dynamics. It's light-induced motion – a touchless motion of droplets."
The research was published in Nature Communications.
Source: MIT








