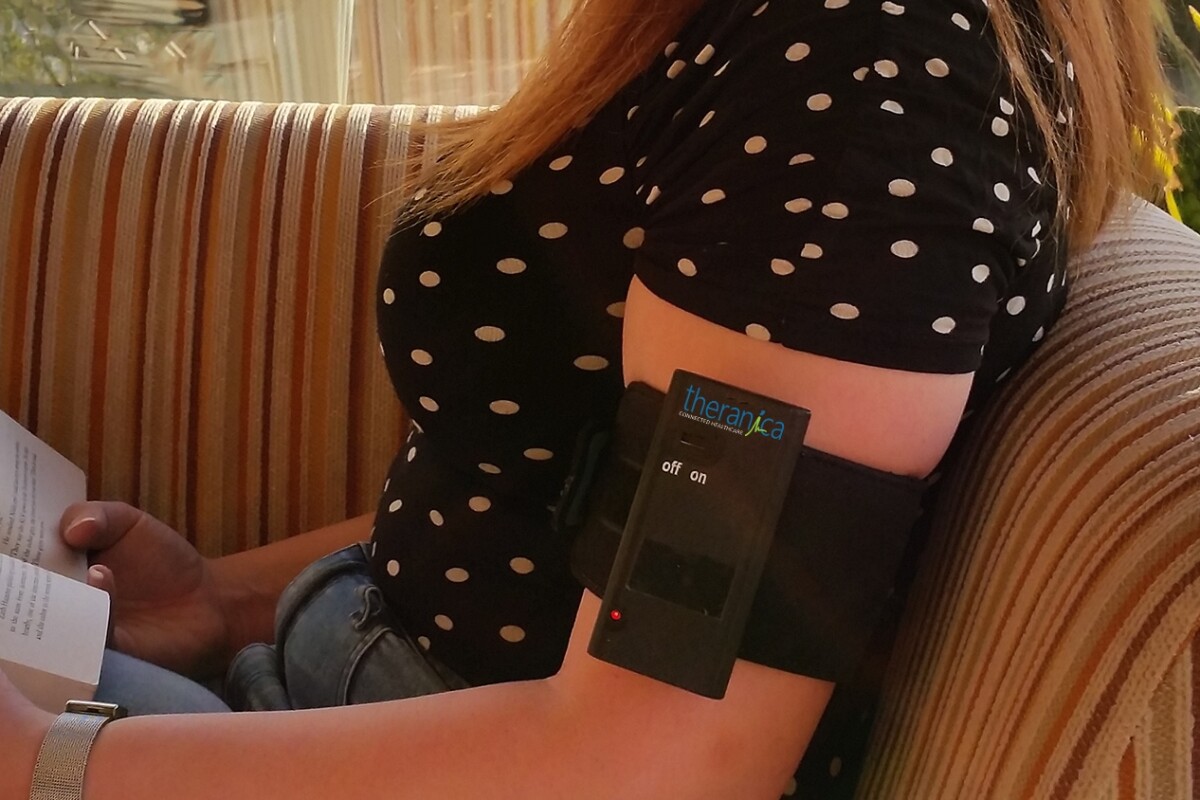
If you suffer from migraine headaches, then you know how awful they can be, potentially disabling their victims for up to 72 hours at a time. Although sufferers generally have to just let them run their course, in recent years we've seen devices such as headbands that are designed to treat them. One of the latest such gadgets – which is worn on the arm like a blood pressure cuff – was recently the subject of a promising study.
Known as Nerivio, the investigational device is made by Israel-based Theranica Ltd.
Among other things, it incorporates a battery, a chip, and two electrodes that make contact with the skin of the upper arm. The company claims that those electrodes deliver electrical pulses which are received by nerve fibers in the arm and relayed up to the brain stem. There, neurotransmitters are activated, which act as natural painkillers.
An app on the user's smartphone is used to wirelessly control the device, allowing users to start treatments as headaches occur, and to adjust the intensity of the pulses – generally speaking, the more intense those pulses are (without actually being painful), the better.
The recent study was carried out by a team led by Dr. David Yarnitsky, of the Technion-Israel Institute of Technology (Yarnitsky is also a member of the medical advisory board for Theranica). It involved 71 test subjects, all of whom suffered from at least eight migraines per month.
Unbeknownst to them, the Nerivio devices they were using were programmed to sometimes deliver levels of stimulation that were strong enough to work for "real" treatments, while at other times producing "sham" treatments that were too weak to be effective. It was found that when the real treatment was delivered, 64 percent of people had a reduction in their pain by at least 50 percent, two hours after the 20-minute treatment period ended – by contrast, only 26 percent of them reported an improvement after the sham. All told, a total of 299 migraines were treated.
According to Yarnitsky, these results are similar to those seen for triptan migraine medications. He does admit, however, that some of the test subjects may have known when they were getting the sham treatments, simply by the weakness of the pulses.
A paper on the research was recently published in the journal Neurology.
Sources: American Academy of Neurology, Theranica