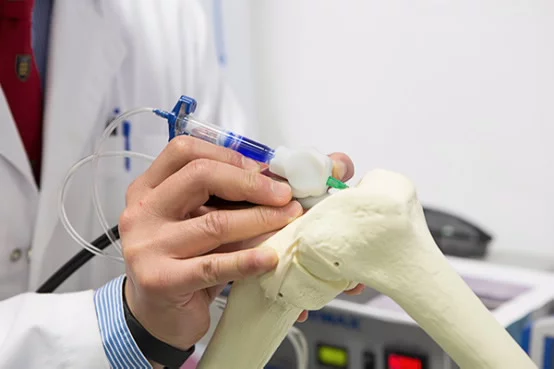
Devices like the 3Doodler and SwissPen literally put 3D printing technology in the hands of consumers, but a new BioPen developed at the University of Wollongong (UOW) in Australia is targeted at more skilled hands. The handheld device is designed to let surgeons "draw" live cells and growth factors directly onto the site of an injury to help accelerate the regeneration of functional bone and cartilage.
Instead of plastic filament, the BioPen extrudes cell material inside a biopolymer such as alginate, which is in turn encased in an outer layer of gel material. Both the outer and inner layers are combined in the pen head as it is extruded and the surgeon "draws" to fill in a section of damaged bone.
The extruded material is solidified as it is dispensed using a low-powered ultraviolet light source fixed to the pen. This provides protection for the embedded cells as the surgeon applies them layer-by-layer to form a 3D scaffold in the damaged bone.

Once the cells have been laid down in the wound site, they multiply and differentiate into nerve, muscle or bone cells to eventually form functioning tissue. As well as cells, the device can also be used to seed growth factors or other drugs to promote regrowth and recovery. The developers say the hand-held design provides both precision in the operating room and makes the device easy to transport.
"This type of treatment may be suitable for repairing acutely damaged bone and cartilage, for example from sporting or motor vehicle injuries," says Professor Choong. "Professor Wallace’s research team brings together the science of stem cells and polymer chemistry to help surgeons design and personalize solutions for reconstructing bone and joint defects in real time.”
The UOW team recently supplied the pen to clinical partners at Melbourne's St Vincent's Hospital, led by Professor Peter Choong, who will work to perfect the cell material for use in clinical trials.
Source: University of Wollongong