A new experimental, non-invasive medical technique is promising to precisely deliver drug-carrying metal nanorods anywhere inside the body and image tissue with cellular resolution. If perfected, the approach could be used to treat inoperable deep-tissue tumors, brain trauma, and vascular or degenerative diseases.
The recent forward strides of nanotechnology are opening the door for medical applications that were hard to fathom even just a few years ago, and span from healing broken bones to precisely delivering drug-carrying nanoparticles to fight cancerous cells.
These techniques rely on a system of externally-applied electromagnets to precisely guide ferromagnetic nanoparticles where they are needed. However, so far the magnets have only been able to attract the particles, not to push them away, making it impossible to reach cells that are located very far from the magnets themselves and deep inside the body.
Researchers at the University of Maryland and Weinberg Medical Physics in Bethesda have now discovered a way to push the nanoparticles away from the magnet, in a crucial advance that opens new possibilities for treating deep-seated inoperable tumors, vascular or degenerative diseases in a non-invasive way.
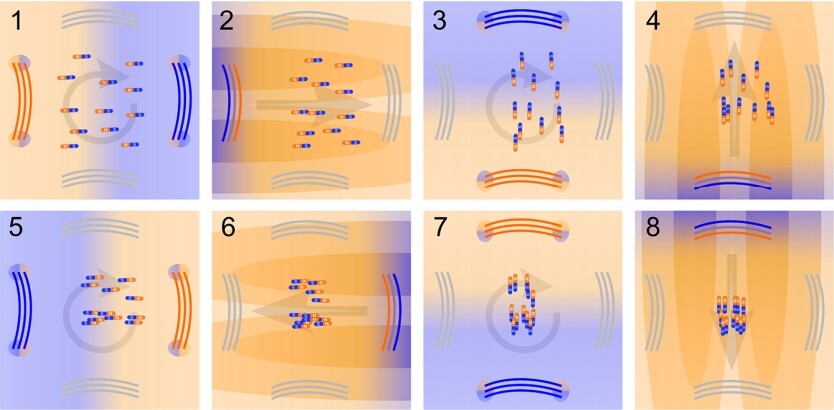
If you’ve ever played with bar magnets, it will be easy to understand why using one magnet to push away another is quite challenging. Like poles repel, but if two magnets with like poles are sitting next to each other, they don’t really tend to stay put: instead, they usually flip in order to align with the existing magnetic field, and then, with their opposite poles next to each other, they naturally attract. Likewise, the external ferromagnets used in nanomedicine inevitably cause the ferromagnetic nanorods to flip and then get closer to the magnet, and are unable to push the rods away toward the deep tissue.
The researchers jumped this hurdle by using quickly-pulsed magnetic fields coordinated in pairs, where the first pulse rotated the metal rods without pulling them and then, crucially, the second pulse followed very shortly after, pushing the rods away from the magnet before the particles had a chance to reorient a second time. By repeating the two steps over four directions, the scientists can precisely control the location of the nanorods and push them further away from the magnets than ever before.
"The new method solves problems common to many types of particle administration, whether intravenous or intra-nasal," Prof. Benjamin Shapiro and Dr. Irving Weinberg told us. "Our particles are designed to do their job (e.g., slowly release drugs, ablate tumors) and then dissolve for eventual excretion in the urine.
"[We] described bare particles that did not carry drugs and could be used to destroy tumors or microbial biofilms, but in other publications we have described the use of ferromagnetic particles as delivery agents, where the payload can be drugs, RNA fragments, or even stem cells."
The researchers used cobalt nanorods in their latest study, but they tell us that the particles can be made out of any ferromagnetic material, including iron or iron oxides. Additionally, the magnetic pulses can not only direct the nanorods, but can also be used to image tissue at the cell level.
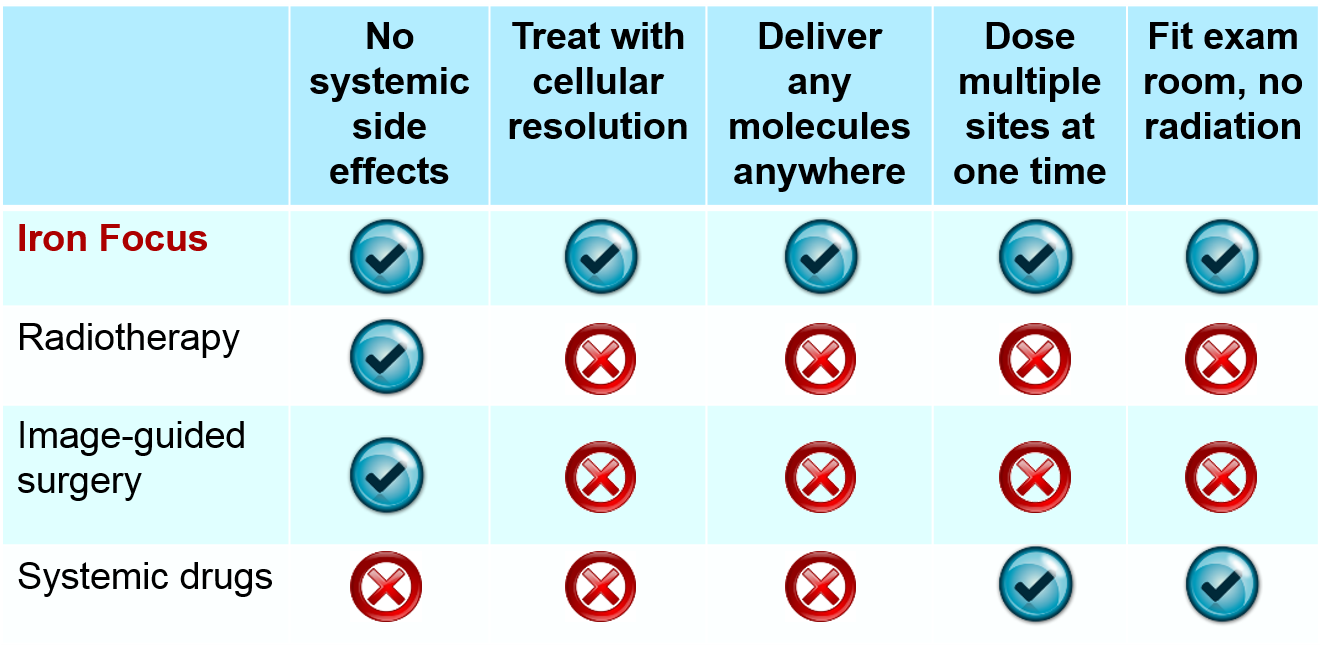
"We have shown that these super-strong [magnetic] gradients (100 times as large as in conventional MRI) are well-tolerated by humans, and can be used to collect MR images with spatial resolution of 15 microns, which is similar to the size of single cells," the researchers told us.
The scientists are now working to demonstrate this method in vivo, and they’re also focusing their efforts on designing their nanoparticles so that they can be mass-produced as easily as possible.
"We are building a combined imaging/targeting platform that will direct particles to foci in preclinical models of infection, neurological disease, and cancer," said the scientists. "Our particles are manufactured with template techniques that is amenable to large-scale production. We are carrying out studies to optimize the nanoparticle design for safe and effective therapy. We are also building spintronic particles that can detect and/or deliver electrical energy once deployed to intended locations."
A paper describing the advance appears in the journal Nano Letters.
The video below demonstrates how the magnetically-controlled nanorods are able to quickly target a precise location.
Source: Iron Focus Medical









