Thomas Edison may have invented the lightbulb, but he never received the Nobel Prize for it. Isamu Akasaki and Hiroshi Amano at the University of Nagoya, and Shuji Nakamura working at Nichia Chemicals in Tokushima, Japan have proven more successful, being awarded the 2014 Nobel Prize in Physics for their invention of the blue LED, which is the key to modern energy-efficient lighting.
The blue light emitting diode (LED) is the basis for modern energy-saving, environmentally friendly light bulbs as well as a range of cutting-edge applications. Invented in its practical form just 20 years ago, the blue LED is the long sought after “missing link" of lighting technology that made LED lamps possible, yet eluded scientists and engineers for 30 years.
For most of human history, lighting was a variation on a lit rag stuck in a bowl of oil, but that changed in 1879 when the Swann-Edison incandescent bulb was invented. This was followed by the first fluorescent light invented by P. Cooper Hewitt in 1900. But until only recently lighting technology has remained an inefficient, energy heavy process.
We think of LEDs as being a modern technology, but the first solid-state light was constructed in 1907 by Henry J Round, and later work was done in the 1920s and ‘30s in the Soviet Union, but a lack of theoretical understanding of the phenomenon hindered any progress and it remained a laboratory curiosity.
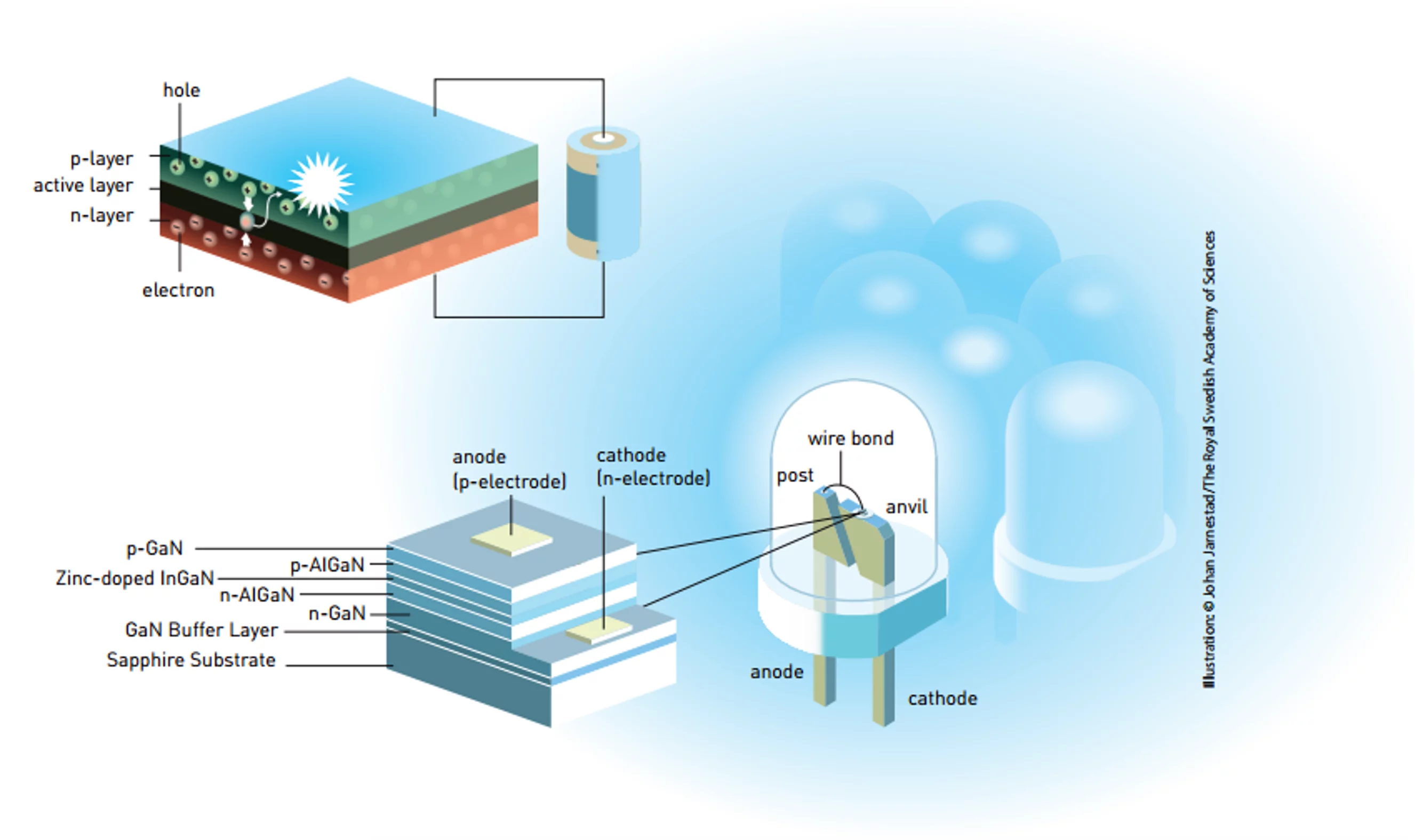
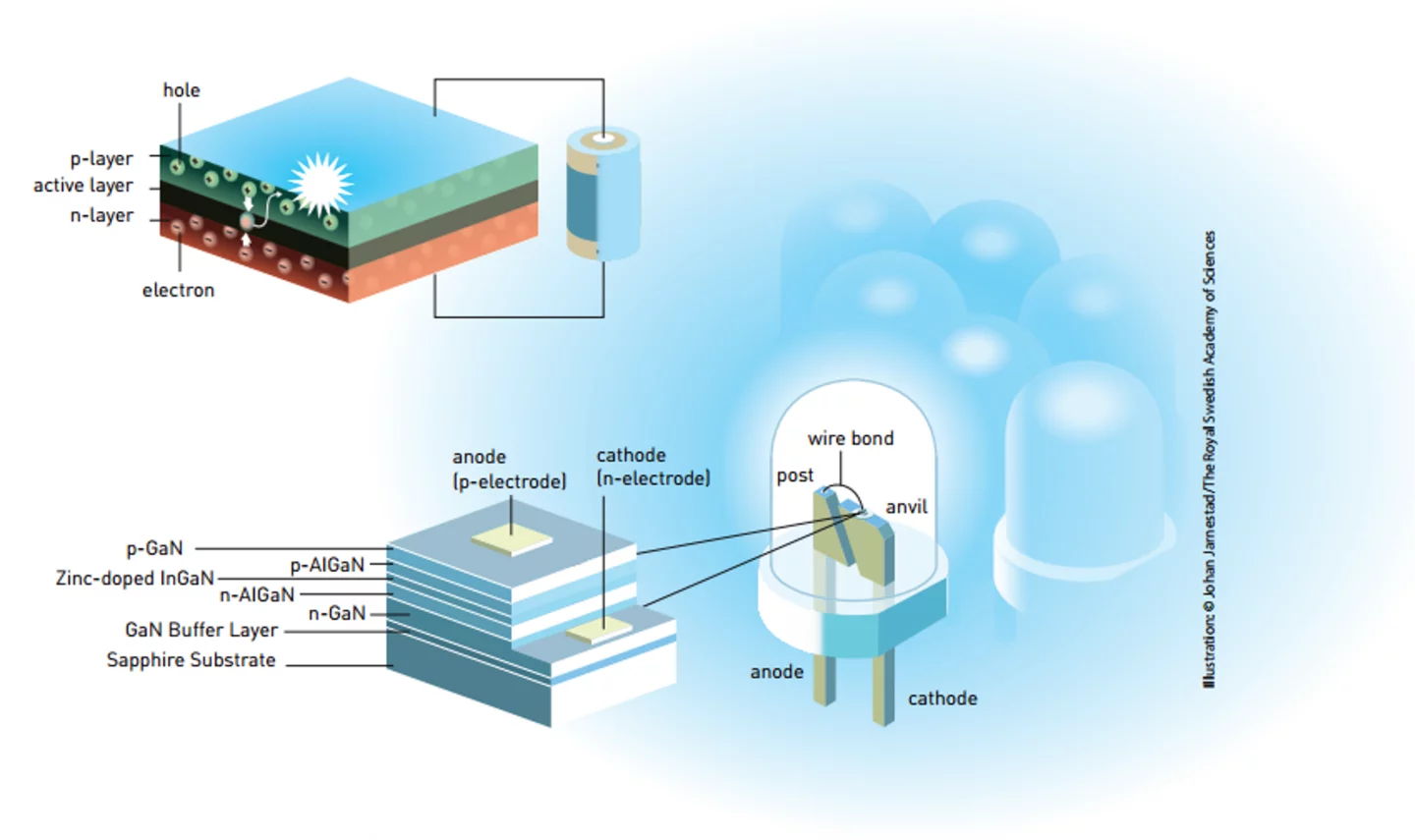
Unlike other light sources, an LED produces light directly instead of generating it has a byproduct of heated filaments or ionizing gas; making it a true “cold” light. It’s composed of layers of semiconducting materials and in its most basic form, it consists of an n-layer, which has an excess of electrons, and a p-layer, which lacks electrons – or could be said to have an excess of positive holes. Between the layers is an active layer. When a current is passed through this, the electrons and holes combine, cancel one another out, and generate light. The wavelength of the light, and hence its color, is based on the materials used.
Thanks to the invention of the transistor and a greater understanding of semiconductor theory, red LEDs were invented in the 1950s and soon found applications as indicator lights and as calculator and digital watch displays. By the late 1960s, green, infrared, and laser LEDs were invented, and LEDs were seen as the future of domestic lighting with popular magazines predicting homes lit by LED panels by 1980.
However, that promise was delayed because, while red and green LEDs were common by 1970, the blue LED needed to create a bright, clean, white light proved much harder to produce. The blue LED was needed to make white light by either working in combination with red and green LEDs, or by shining through a phosphor that creates red and green light. However, it was a classic case of “first catch your rabbit” that required a long series of breakthroughs in basic materials physics and crystal growing.
Despite work since the late 1950s, finding the right material to create the sought-after LED was elusive. Either the material was theoretically right, but could be grown as little more than dust particles, or could be formed into crystals, but weren't up to the job.
Akasaki, Amano, and Nakamur, worked on their two separate efforts for years on the problem – often building their own equipment. The key was gallium nitride, which theoretically could be used to create a blue LED, but which in practice proved to be very uncooperative. The metallic crystals needed to be of extremely high quality and the alloy was very prone to poisoning by hydrogen, so making p-layers of adequate purity was not easy, and the use of electron beams to make the diodes was difficult and expensive.

By 1986, Akasaki and Amano produced a gallium nitride crystal on a layer of aluminum nitride on a sapphire substrate, and a few years later managed to make a p-layer. In 1992, they built their first LED light. Meanwhile, Nakamura created his own light two years later using a more efficient heating process that drove off the hydrogen as effectively as an electron beam. This was followed by improvements by both teams as they explored new gallium nitride alloys using aluminum or indium, as well as more complex diode designs.
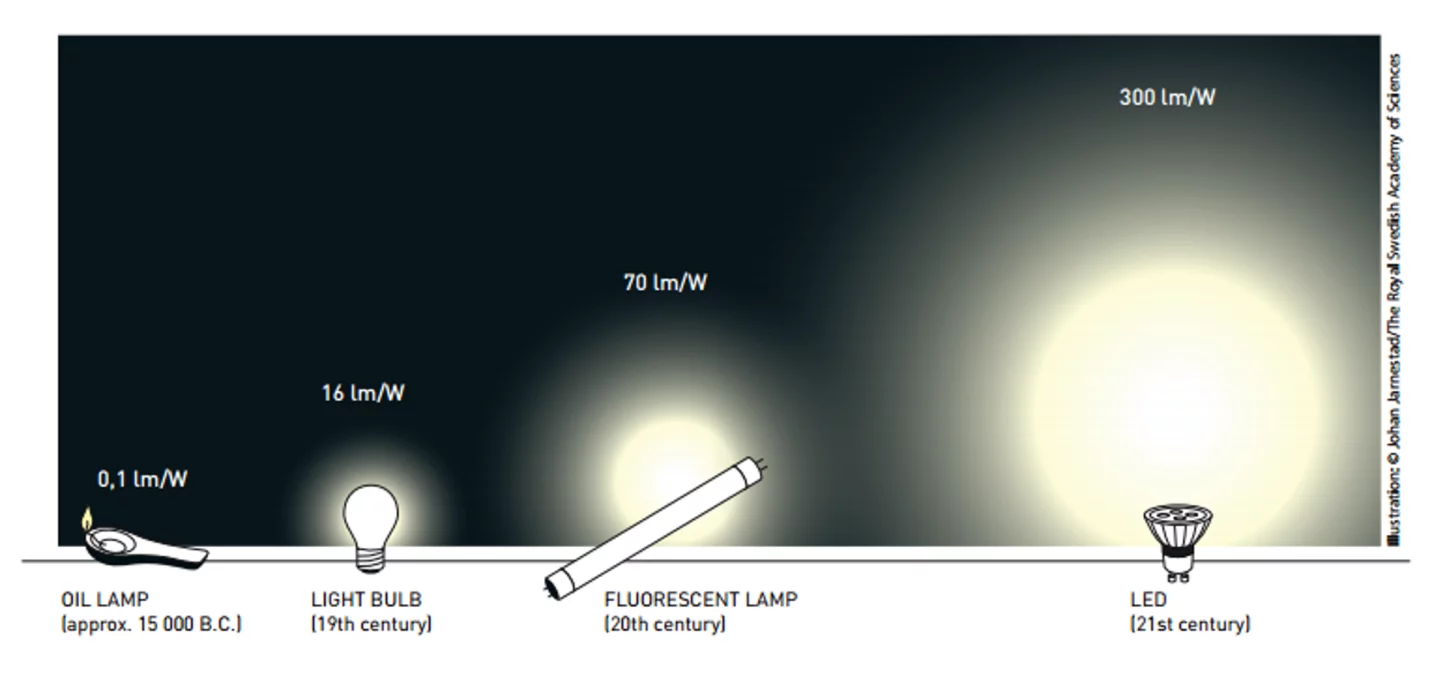
The impact of this invention would be hard to overestimate. LED lamps give out light at up to 300 lumens per watt (lm/W) compared to 16 lm/W for conventional incandescent bulbs and 70 lm/W for fluorescents. Aside from domestic and commercial lighting, they’re used in car headlamps, yard lights, Christmas lights, camera flashes, and pocket torches. In addition, the technology has allowed the development of LCD televisions, LED lasers for Blu-ray players and more efficient laser printers, as well as ultraviolet LEDs, which are used for everything from sterilizing bottled water to combating the spread of the Ebola virus.
Along with their practical applications, blue LEDs have had a beneficial impact on the environment. They’re made without the controversial mercury found in compact fluorescent bulbs, and with a quarter of the world’s energy output going to lighting, the cold light of the LED has a great potential for reducing demand on energy grids. In addition, LEDs last up to 100,000 hours instead of the 1,000 hours for incandescents and 10,000 hours for fluorescent lamps. Also, like mobile phones, they're an example of a technology that can easily be adopted in less developed countries in the form of self-contained solar-powered lamps.
According to the Nobel Foundation, blue LEDs still have a long way to go as the technology matures with more efficient, adjustable white lights based on a triad of advanced red, blue, and green LEDs coming on line. Eventually, the Foundation sees large, computer-controlled LED panels covering hundreds of square yards that can change colors or patterns, that can adjust lighting to suit natural biorhythms, or provide more efficient greenhouse lighting.
Akasaki, Amano and Nakamura will receive their prizes at a ceremony in Stockholm in December.
Source: The Nobel Foundation