Once released into the environment from industrial sources, trace amounts of heavy metals can remain present in waterways for decades or even centuries, in concentrations that are still high enough to pose a health risk. While processes do exist for removing larger amounts of heavy metals from water, these do not work on smaller quantities. Now, however, scientists from Rhode Island's Brown University have combined two existing methods, to create a new one that removes even trace amounts of heavy metal from water.
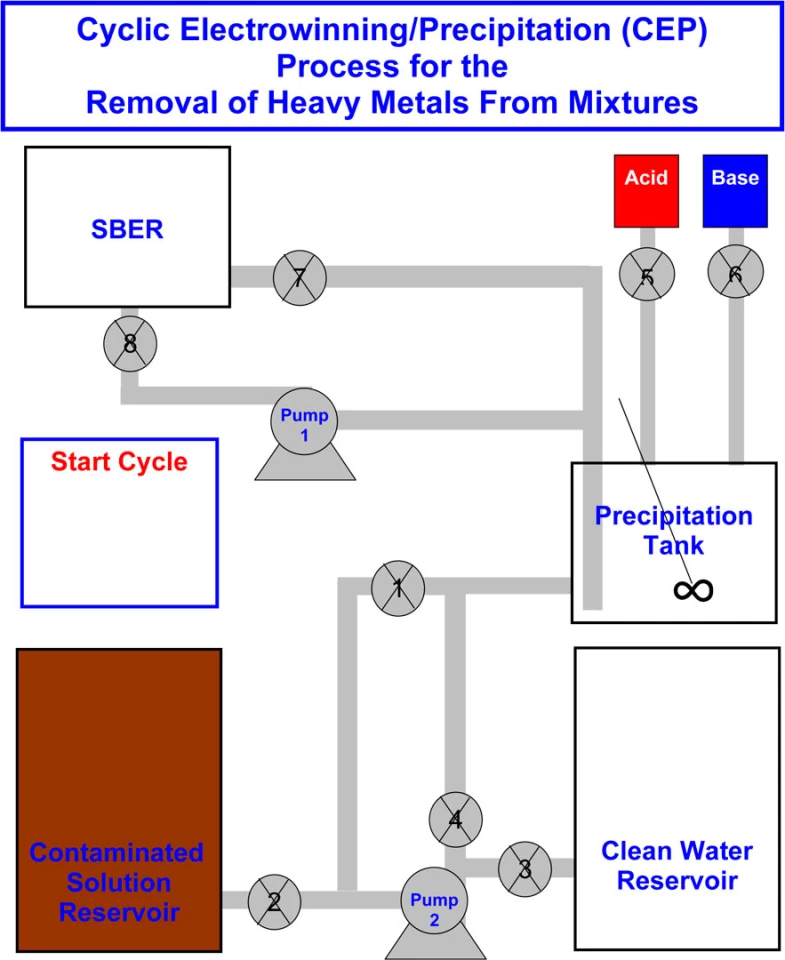
Known as the cyclic electrowinning/precipitation (CEP) system, the process involves increasing the concentration of heavy metals in water samples, until it's high enough to be effectively removed.
Things get started when metal-tainted water is fed into a tank, and an acid or base (such as sodium hydroxide) is added to change the water's pH value. This causes the water molecules to separate from the heavy metal precipitate, which settles to the bottom of the tank. The clean water is then siphoned off, more tainted water is introduced, and its metal content joins that already lying at the bottom. The process is repeated a number of times.
Although this technique alone holds promise, the settled precipitate forms into a toxic sludge, which is difficult to safely dispose of. That's where part two of CEP comes into play.
Once the sludge in the first tank has reached a high enough heavy metal concentration, it is pumped into a second device called a spouted particulate electrode. There, it is subjected to a process called electrowinning, in which an electrical current is used to transform the metal ions into a stable, solid, and thus easily-removed state. The water left over is then returned to the first tank, where most of the remaining metal content is settled out. That water then goes to another reservoir, where other processes are employed to further remove heavy metal ions.
In tests of the system, trace amounts of heavy metals such as cadmium, copper, and nickel were removed from tainted water samples, to the point that the water ended up near or below the Environmental Protection Agency's maximum contaminant levels. Project leader Prof. Joseph Calo says that CEP could also be used to remove metals including lead, mercury, and tin.
A paper on the Brown University research was recently published in the Chemical Engineering Journal.





