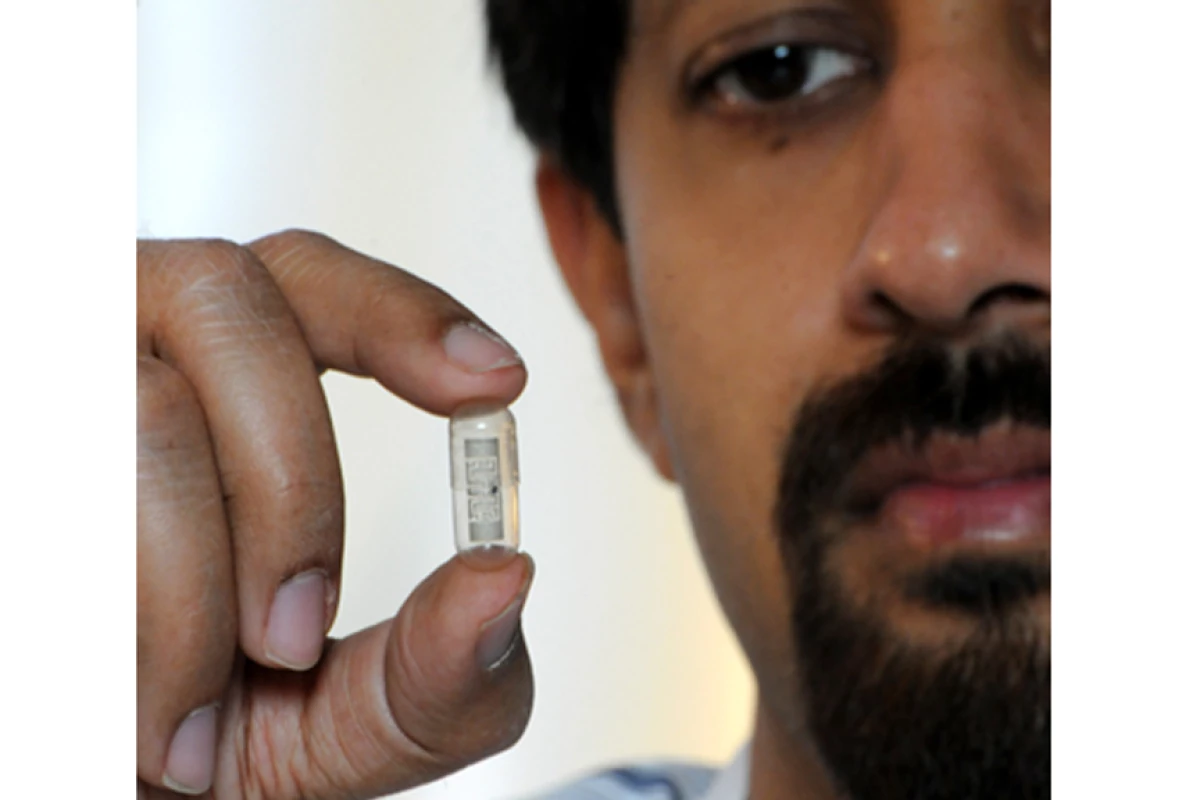
Patients forgetting, bungling or just plain refusing to take their medication is a big problem for health care professionals and patients alike. It can exacerbate medical problems, spurring hospitalizations or expensive medical procedures and undercut clinical trials of new drugs. In seeking a way to confirm that patients have taken their medication a team of researchers have added a tiny microchip and digestible antenna to a standard pill capsule that automatically alerts doctors when the pill has actually been ingested.
The system developed by a team at the University of Florida (UF) consists of two main parts. The first is the pill, which is a standard white capsule coated with a label embossed with silvery lines. These lines comprise the antenna, which is printed using ink made of nontoxic, conductive silver nanoparticles. The pill also contains a tiny microchip about the size of a period.
When a patient takes a pill, it communicates with the second main element of the system - a small electronic device carried or worn by the patient. At the moment this is a stand-alone device, but in the future the researchers say it could be built into a watch or mobile phone. The device then signals a cell phone or laptop that the pill has been ingested, in turn informing doctors or family members.
The pill doesn’t require a battery as the stand-alone device sends it power via imperceptible bursts of extremely low-voltage electricity. The bursts energize the chip to send signals relayed via the antenna. Eventually the patient’s stomach acid breaks down the antenna and the microchip is passed through the gastrointestinal tract - but not before the pill confirms its own ingestion.
The team has successfully tested the pill system in artificial human models, as well as cadavers. Researchers have also simulated stomach acids to break down the antenna to learn what traces it leaves behind. According to Rizwan Bashirullah, UF assistant professor in electrical and computer engineering, those tests had determined the amount of silver retained in the body is tiny, less than what people often receive from common tap water.
The prototype is intended to pave the way for mass-produced pills that will address the failure of patients to follow prescription regimens – which the American Heart Association cites as “the number one problem in treating illness today.” Studies have found, for example, that patients with chronic diseases normally take only about half their prescribed medications. According to the American Heart Association, 10 percent of hospital admissions result from patients not following the guidelines on their prescriptions. Other studies have found that not taking medication properly results in 218,000 deaths annually.
So-called “medication compliance” is a big problem for clinical trials, Bashirullah said, because failure to take experiment drugs skews studies’ results or renders them meaningless. As a result, researchers often require visual confirmation of participants taking pills, an extremely expensive proposition if hundreds or thousands of people are participating in the trials.
“The idea is to use technology to do this in a more seamless, much less expensive way,” Bashirullah said.
The researchers are currently working on a scholarly paper about their research. They have also applied for patents, and Bashirullah said a UF spinoff company is seeking to develop the next generation of the pill for FDA testing and commercial development.