The wonders of graphene seem to know no bounds. Not only is it one of the strongest materials known, is both highly conductive and piezoelectric, it can generate electricity from flowing water and now it is being used to make better supercapacitors. Using a DVD writer, a team of UCLA researchers has invented a new process for making high quality graphene electrodes and used these electrodes to make a new species of supercapacitor. Though the work is in the early stages of development, it could lay a foundation for supercapacitor-based energy storage systems suitable for flexible portable electronic devices.
Lithium-ion batteries are electrically fragile, can explode on charging, and must be slowly recharged over a period of hours to avoid an early death. Supercapacitors, more formally known as electric double layer capacitors, are rugged and can be charged in a minute or so. They also can provide plenty of power and last through millions of recharge cycles. Why, then, don't we hear more about their use?
The short answer is energy density. Supercapacitors store about 20 watt-hours per kilogram, or one-seventh of the energy per kilogram of a lithium-ion battery.
To understand what more widespread adoption will require, let's take a look at how supercapacitors work. A supercapacitor is able to store a charge as a coating of ions adsorbed on the surfaces of its electrodes.

Ions are separated from the electrolyte by a charging current, and are propelled toward their respective electrodes. The membrane serves to separate the ions so that a net charge separation can be maintained. Notice that the ions on the electrode surface are neutralized (save for a residual dipole field) by the opposite charge attracted to just below the surface of the electrode. This dual surface layer is called an electric double layer. All other parameters being equal, the number of ions stored is proportional to the surface area of the electrodes. Energy storage of a capacitor is proportional to the amount of charge stored, so a tenfold increase in capacitance will require new electrodes that are highly conductive (so large power levels can be generated) and provide more surface area than conventional supercapacitors.
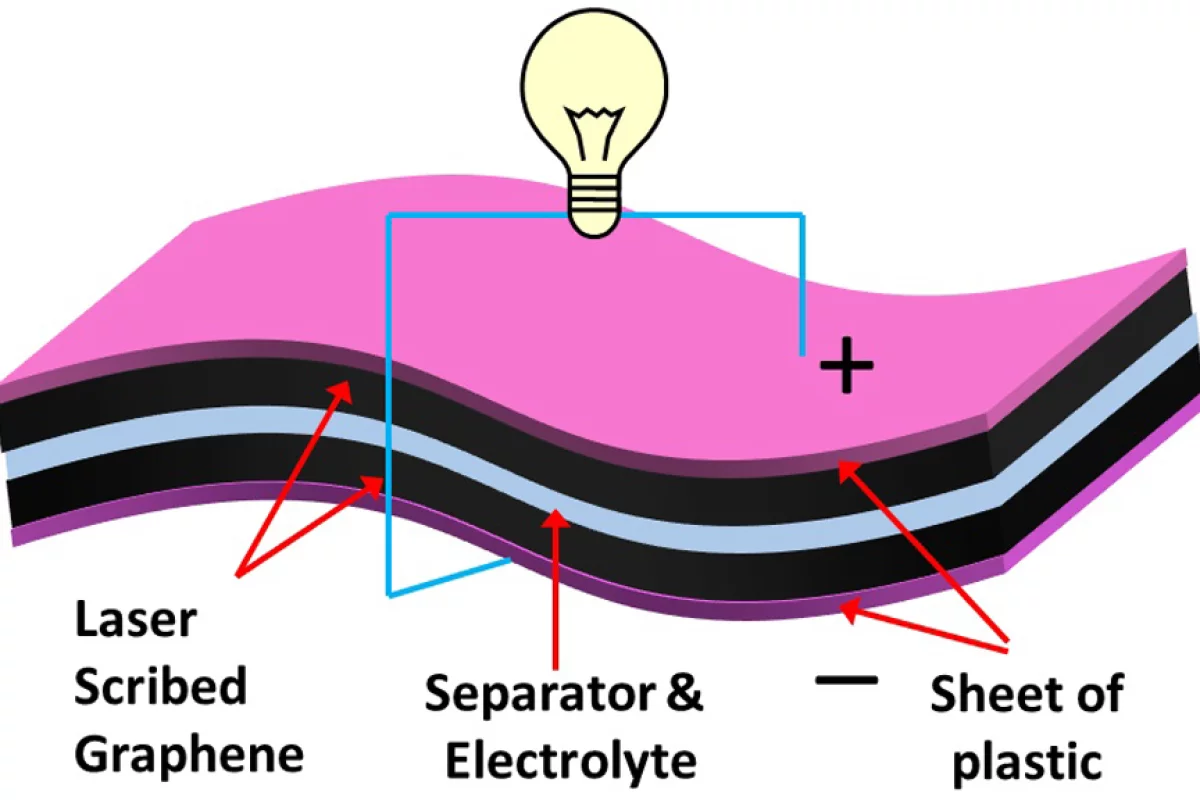
Now researchers at UCLA have used a standard DVD writer to make such electrodes. The electrodes are composed of an expanded network of graphene that shows excellent mechanical and electrical properties as well as exceptionally high surface area.
The process is based on coating a DVD disc with a film of graphite oxide that is then laser treated inside a DVD writer to produce high-quality graphene electrodes. Graphite oxide is a compound of carbon, oxygen, and hydrogen made by treating graphite with sulfuric and phosphoric acids combined with potassium permanganate, an extremely strong oxidizer. When graphite oxide is placed in a basic solution, it exfoliates into monomolecular layers with a graphene-like structure. These layers were then collected on an ordinary DVD disc. The disk was then written on, a number of passes being made.
The action of the 5 milliwatt IR laser on the graphite oxide was to reduce the material, thereby producing isolated but intertangled graphene monolayers. The surface area of the resulting electrodes was 1,520 square meters per gram - about a third of an acre, and 3-5 times the surface area of activated carbon electrodes. You can't do much better, as graphene's intrinsic surface area is 2,630 square meters (about two thirds of an acre) per gram. Also of importance is the conductivity of the electrode material - at 10-100 times that of activated carbon and previous graphene electrodes, we are assured that high power densities are at hand.
The UCLA research team investigated several different types of supercapacitor chemistry using the laser scribed graphene electrodes. They found the supercapacitors were surprisingly robust against flexure, surviving thousands of folds with no significant change in capacitance. Their highest energy storage supercapacitor was based on using the ionic liquid 1-ethyl-3-methylimidazolium tetrafluoroborate as the electrolyte. The supercapacitor exhibited a capacitance of 276 Farads per gram, and an operating voltage of 4 volts. This corresponds to an energy density of over 600 watt-hours per kilogram (2.2 lb), or about four times that of lithium-ion batteries. In practice, the energy density will be smaller, owing to support structures, but such supercapacitors should be able to give lithium-ion batteries a run for their money.
The UCLA team's paper is published in the journal Science.
Source: UCLA