A team at Stanford claims to have made a battery breakthrough that could boost the performance of lithium-ion batteries and also make them smaller and lighter. The researchers managed to remove two long-standing barriers to these improvements by putting silicon particles in graphene "cages."
To improve capacity in recent years batteries have begun to use silicon anodes, which have more capacity than the graphite conventionally used. But silicon particles also swell so much during charging that they're prone to cracking or shattering and they can also react with the battery electrolyte, forming a coating that reduces performance.
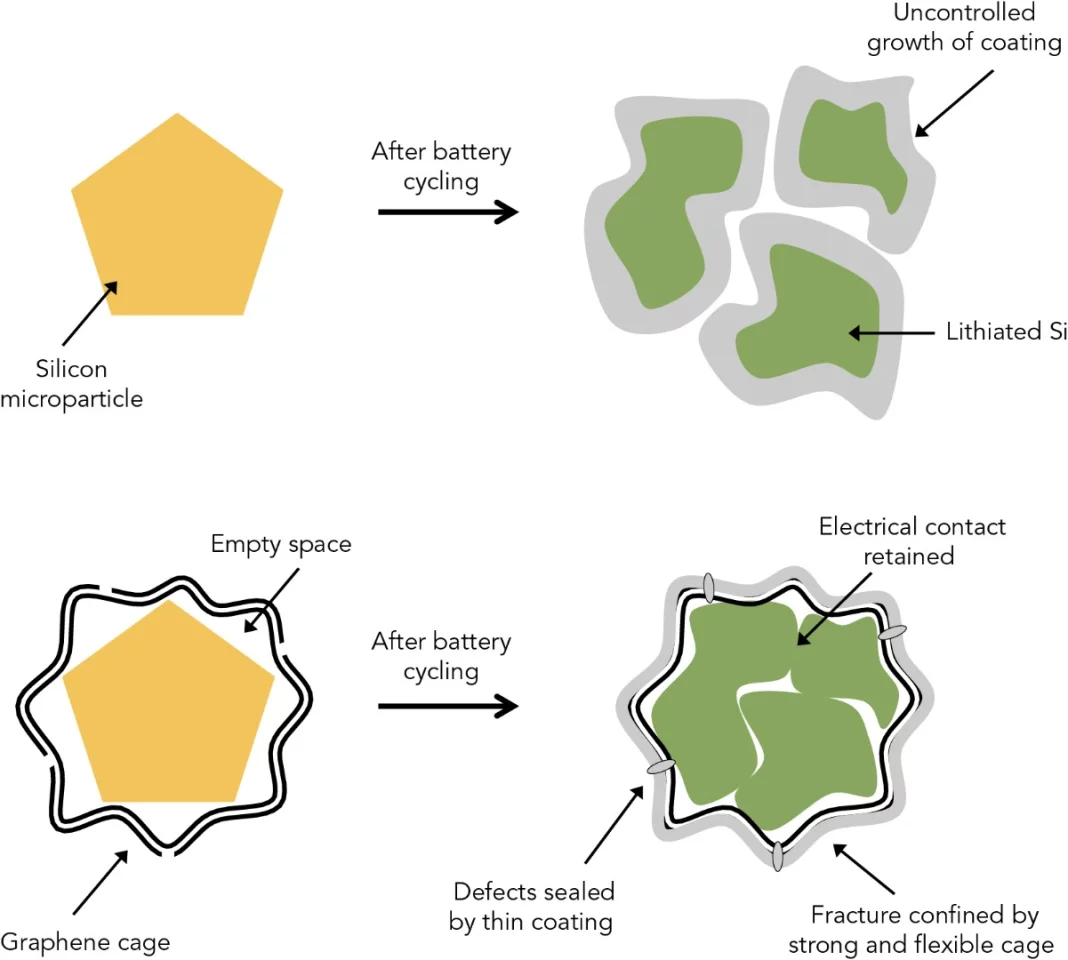
The solution from the team at Stanford and the Department of Energy's SLAC National Accelerator Laboratory is to encase each silicon particle in a "custom-fit cage" of graphene. At only one-atom thick, graphene is the thinnest, strongest form of carbon and also conducts electricity well.
The carbon cages would allow the silicon to expand and even break apart, but keep the pieces together so that they can continue to function. The graphene barrier would also block the destructive chemical reactions with the electrolyte from occurring.
"In testing, the graphene cages actually enhanced the electrical conductivity of the particles and provided high charge capacity, chemical stability and efficiency," said Yi Cui, an associate professor at SLAC and Stanford who led the research. "The method can be applied to other electrode materials, too, making energy-dense, low-cost battery materials a realistic possibility."
Cui's team first attempted a similar approach using expensive nanoparticles and different coatings, but were not able to achieve a high enough level of efficiency.
"This new method allows us to use much larger silicon particles that are one to three microns, or millionths of a meter, in diameter, which are cheap and widely available," Cui said. "In fact, the particles we used are very similar to the waste created by milling silicon ingots to make semiconductor chips; they're like bits of sawdust of all shapes and sizes. Particles this big have never performed well in battery anodes before, so this is a very exciting new achievement, and we think it offers a practical solution."
Getting a form-fitting coat of graphene on each silicon particle is a bit of process. The particles were first coated with nickel, and layers of graphene were then grown on top of the metal. Acid is then used to dissolve the nickel, leaving enough of a void within the cage for the silicon to expand.
"The form-fitting graphene cages are the first coating that maintains high efficiency, and the reactions can be carried out at relatively low temperatures," said Stanford postdoctoral researcher Kai Yan.
Next the team will hone the process to produce large enough quantities of the caged particles to create prototype commercial-scale batteries.
The work is described in the most recent issue of Nature Energy.
Source: Stanford





