Lithium is expensive, environmentally questionable in large volumes, and tends to catch on fire from time to time. It's the best solution we've currently got for EV and device battery storage, but other alternatives are starting to surface, and one that could actually make a fair bit of sense is the potassium metal battery.
As well as being cheap and abundant, potassium tends to be easier to work with, meaning manufacturing costs are lower as well as material costs. Using full potassium metal anodes, batteries can be built with energy densities (both by volume and weight) that are comparable to what lithium offers.
The problem up until now has been one that's been present in lithium batteries as well: dendrite formation. Over time, as the battery is charged and discharged again and again, bits of the metal – ether lithium or potassium – start to attach themselves to the anode. This doesn't happen evenly; little spiky branches called dendrites begin to form, and eventually, they get long enough to poke through the insulating membrane separating the anode from the cathode, and they short circuit the battery. This causes heat build-up and occasionally fires, and effectively reduces the lifespan of a battery.
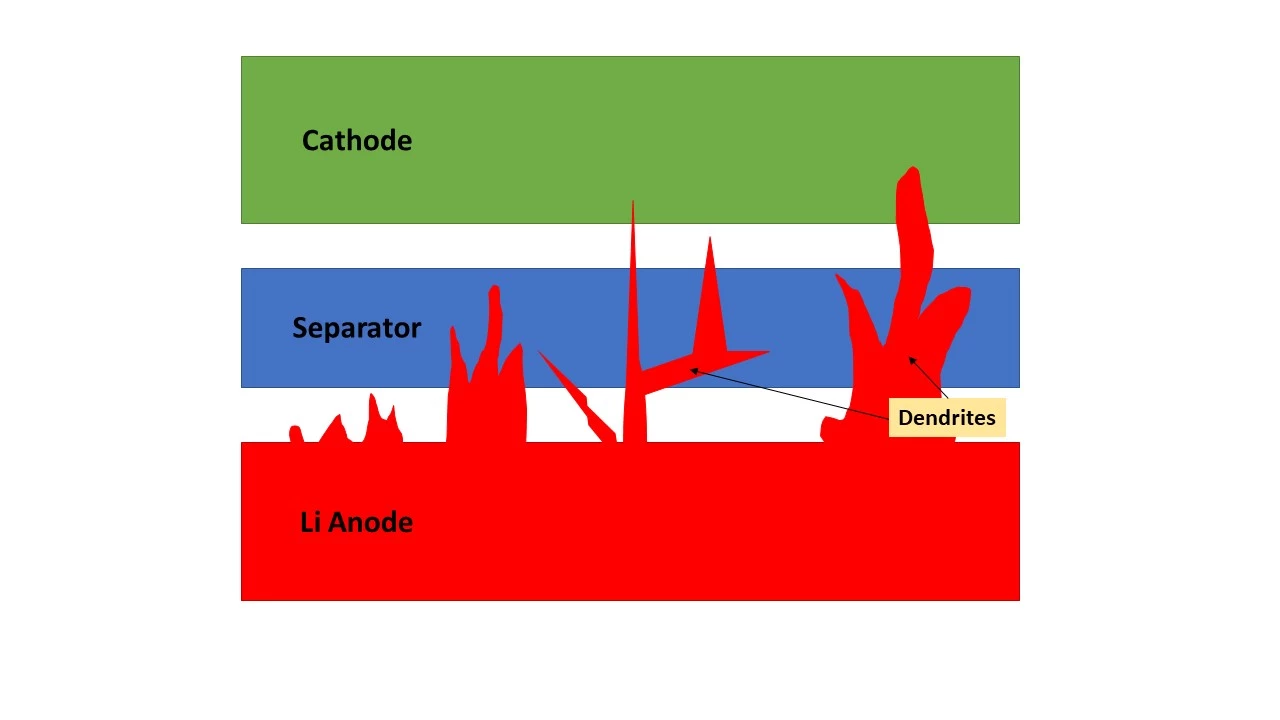
Now, a team of scientists from the Rensselaer Polytechnic Institute in New York state say they've developed a self-healing technique that can clean these dendrites off the anode as you charge the battery overnight, preventing them from ever getting long enough to cause problems. They believe it's now possible to bring cheap, long-lifespan potassium metal batteries into mass-market use.
The technique sounds odd, because it's essentially blast-charging and discharging at a high rate; exactly what we're told we shouldn't do if we want to extend the life of our lithium batteries. By doing so under controlled circumstances, the researchers say they can precisely control the heat in the battery, raising it to the point where the potassium doesn't melt, but it activates surface diffusion in such a way as to shift the metal sideways off the dendrite pile and smooth it out.
The same research team demonstrated a similar self-healing capability for dendrites in lithium batteries back in 2018, but it required significantly higher temperatures.
"With this approach, the idea is that at night or whenever you're not using the battery, you would have a battery management system that would apply this local heat that would cause the dendrites to self-heal," said Nikhil Koratkar, an endowed professor of mechanical, aerospace, and nuclear engineering at Rensselaer and the lead author on this research. "I want to see a paradigm shift to metal batteries. Metal batteries are the most efficient way to construct a battery; however, because of this dendrite problem they have not been feasible. With potassium, I'm more hopeful."
The study was published in PNAS.
Source: Rensselaer Polytechnic Institute






