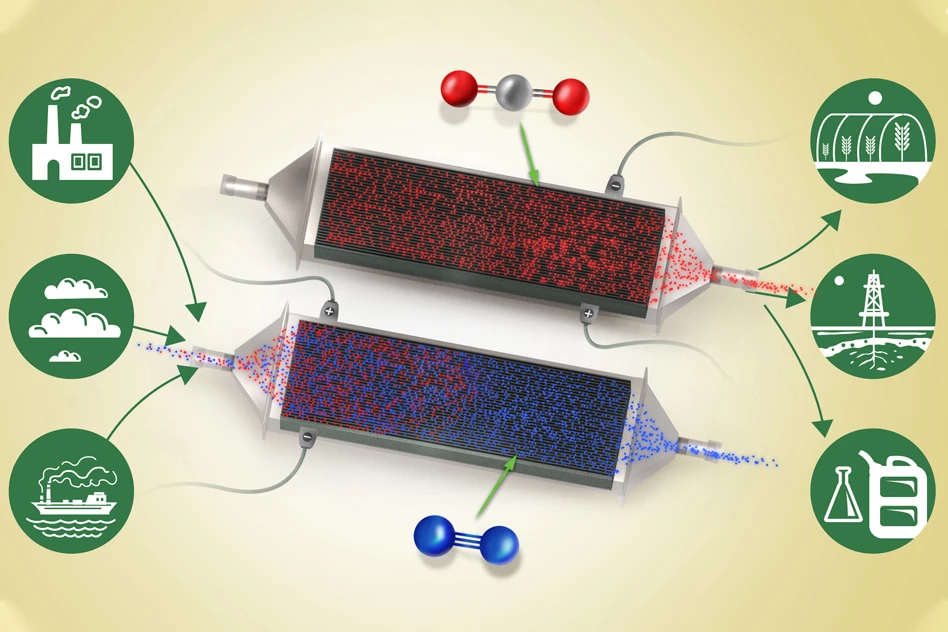
Researchers at MIT have developed a new method for pulling carbon dioxide out of any passing gas stream – whether that’s a chimney flue at a power plant or even just ambient air. The new device is basically a specialized battery that can absorb carbon dioxide while it’s charging, then release it as a pure, steady stream for industrial use when discharging.
The new device is made up of chambers that hold an array of battery electrodes, arranged in stacks with small gaps between them that allow gas to flow through. These electrodes are coated in a compound called polyanthraquinone, which is made up of carbon nanotubes.
When the battery is charging, an electrochemical reaction takes place at the surface of each electrode stack. Because the electrodes have a natural affinity for carbon dioxide, molecules of it floating past will be attracted to them, sticking to the walls of the electrode.
When those electrodes are too full of CO2 to hold any more, the system can then be run in reverse to clear it out. Discharging the battery will detach the molecules from the walls, where they can then be blown out into a separate chamber as pure CO2, which itself is a useful industrial product.
The team says that the most useful aspect of the device’s design is that the electrodes’ affinity for carbon dioxide is essentially all or nothing – when it’s charging, it has a high affinity for the molecule, but while discharging it drops to basically zero, efficiently clearing out the captured carbon.
“The greatest advantage of this technology over most other carbon capture or carbon absorbing technologies is the binary nature of the adsorbent’s affinity to carbon dioxide,” says Sahag Voskian, co-lead author of the study. “This binary affinity allows capture of carbon dioxide from any concentration, including 400 parts per million (the levels in the atmosphere), and allows its release into any carrier stream, including 100 percent CO2.”

The researchers say that the system could be scaled up relatively easily, and outlined how it might be put to work in, for example, a power plant setting. Two sets of these electrode stacks could be placed in a flue, with one charging and one discharging at any given time. Exhaust gas is directed into the charging electrode stacks to capture CO2, while the others are discharging and releasing the captured carbon into a separate chamber. When the charging stacks are full, the process is reversed, letting them empty their CO2 cache while the gas is diverted to the freshly-discharged stacks.
That process would not only drastically reduce the CO2 pumped into the atmosphere, but it would produce a steady stream of pure CO2 gas. This can then be used in applications such as carbonating drinks or feeding plants in greenhouses, which normally have to make it from scratch, often by burning fossil fuels. Otherwise the gas could be sequestered straight underground, or processed into other fuels.
The team says that the device is relatively energy efficient, using up about one gigajoule of energy per ton of carbon dioxide captured. Others, they say, can consume between one and 10 gigajoules per ton. Better yet, other similar systems usually require more energy like heat, or chemical processing to release the captured carbon.
In lab tests, the researchers showed that it could endure 7,000 charging and discharging cycles, with a 30 percent reduction in efficiency. In the future the team believes that it can be bumped up to 20,000 to 50,000 cycles.
The research was published in the journal Energy and Environmental Science. The device is demonstrated in detail in the video below.
Source: MIT