Red wine and chocolate are notorious for staining clothes, thanks to the tannic acid that they and other substances contain. Scientists at the University of Tokyo have now used that acid in a textile coating which keeps clothes from stinking, and that doesn't wash out.
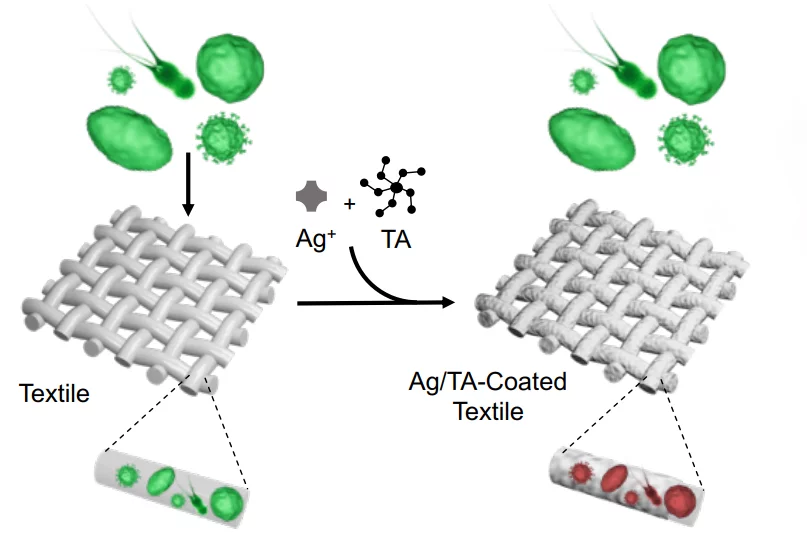
Along with the tannic acid (which is a type of polyphenol), the ultra-thin coating also incorporates silver nanoparticles.
While silver is well known for its ability to kill odor-causing bacteria, researchers have previously had difficulty chemically binding it to fabric. This is an important consideration, not only because frequently reapplying a silver coating would be a hassle, but also because silver nanoparticles may pose a health hazard when released into the environment.
That's where the new Ag/TA (silver/tannic acid) coating comes in.
The tannic acid binds with both the silver and the fabric, keeping the two securely bound to one another. And although tannic acid does produce colored stains in large amounts, the coating itself is colorless. It could be applied to fabrics in either of two ways – textile manufacturers could dip sheets of material in an Ag/TA solution, or consumers could spray it onto items of clothing themselves.
In tests conducted so far on cotton, polyester and silk, the coating has proven to be highly effective at killing not only bacteria but also fungi and viruses. What's more, it has maintained that functionality for at least 10 wash cycles, plus the application of the coating didn't affect the materials' flexibility or breathability.
"This isn’t just a hypothetical situation limited to the lab, I’ve tried it on my own shirts, socks, shoes, even my bathmat," said postdoctoral fellow Joseph Richardson, who is leading the study along with Assoc. Prof. Hirotaka Ejima. "We’d like to see what other useful compounds polyphenols might help bind to fabrics. Antimicrobial silver might just be the start."
A paper on the research was recently published in the journal Scientific Reports.
Source: University of Tokyo