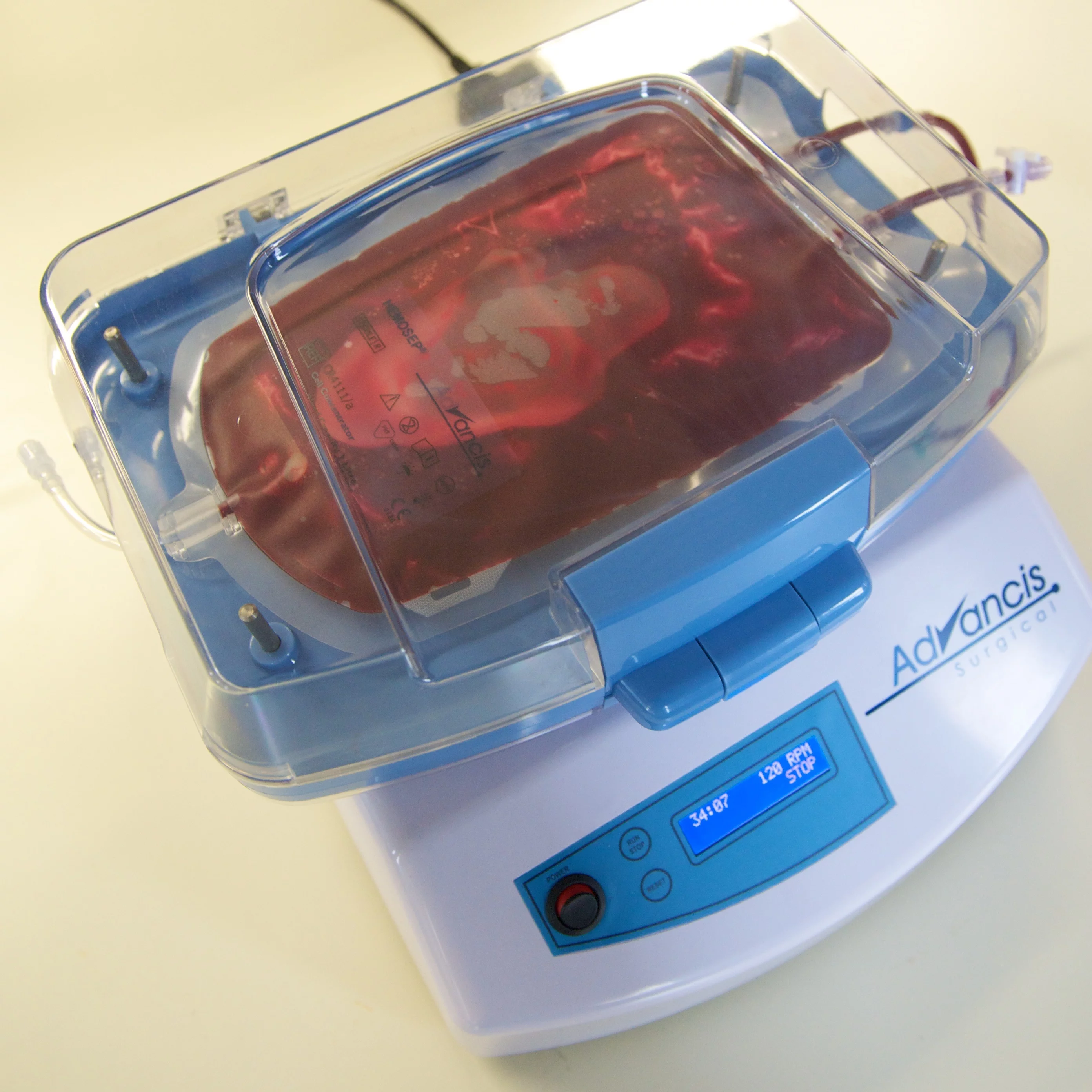
A simpler, cheaper and more efficient method to recover blood lost during open heart and major trauma surgeries has been developed at the University of Strathclyde in Glasgow. Called HemoSep, the new approach has proven successful improving prospects for cardiac surgery patients during trials at the Kirikkale University Hospital in Ankara, Turkey. The technology is soon to be launched commercially.
HemoSep is an autotransfusion method, which concentrates cells from the blood spilled during surgery and recovers them for transfusion back to the patient. This is done with the help of blood bag that employs a chemical sponge technology. A mechanical agitator does the job of concentrating the blood taken from the site of the surgery or drained from the heart-lung machine after the surgery is finished. Finally, the patient receives the recovered blood through intravenous transfusion.
The clinical trials consisted of over 100 surgical procedures and HemoSep significantly reduced the need for blood transfusions, besides preserving normal clotting mechanisms. On top of that, the occurrence of inflammation, a common reaction after such procedures, was also reduced.
This method offers several advantages over traditional ones, mainly because it reduces need for donor blood and the risk of adverse reactions during transfusion, such as contamination and immunosuppression. It also simplifies the process. Traditional donor blood methods can be complex and costly, requiring a sophisticated centrifuge and a pumping machine that has to be operated by specialized technicians.
The team that developed the new method expects the technology will be adapted for open-heart surgery on children, who face greater risks during such procedures. They are positive the method could become standard during heart surgeries and there is also a possibility that it could be applied to blood preservation in cases of transplantation, orthopedics and neurosurgery.
HemoSep has received a CE mark, which means it can be sold to the health sector. The technology has been licensed to Advancis Surgical Ltd., and the company will commercialize the technology in the European Union, Canada, where it received national approval, and other regions that recognize the CE mark. The launch of the device is scheduled for September and further trials are also planned.
Source: University of Strathclyde