Aselectronics miniaturization heads towards a theoretical physical limit in thetens of nanometers, new methods of manufacturing are required to producetransistors, diodes, and other fundamental electronic components. In this vein, a new range of molecule-sizeddevices have been created in the laboratory, though with varying results interms of efficiency and practicality. Now a group of researchers from BerkeleyLab and Columbia University claims to have created the highest-performing,single-molecule diode ever made, which is said to be 50 times better inperformance and efficiency than anything previously produced.
Ordinarydiodes are usually constructed from silicon with a p-n (positive-negative)junction created at the point of contact between a positively "doped" semiconductor (that is, one that has had its electrical properties altered with additives) and a negatively doped one. Flanked by connecting electrodes (an anode on one side and a cathode on the other), the most common function of such a diode is to permit electric current to flow in onedirection only, whilst blocking current from flowing in the reverse direction. Assuch, a diode used in this way can be seen as a type of flow-control valve thatis either "on" or "off". Technically, this one-way behavior is known as rectificationas it can, for example, be used to rectify alternating current to direct current, and so thesetypes of diodes are known as rectifiers.
This on/off – asymmetric – behavior in the nascent fieldof molecule-sized electronics, on the other hand, is usually achieved by the creation of moleculesthat chemically emulate the p-n junction. However, these synthesized molecular junctionshave generally resulted in poor forward current flow capabilities and inefficientor patchy rectification. This is where the Columbia university scientists claimto have made significant improvements with their new single-molecule diode.
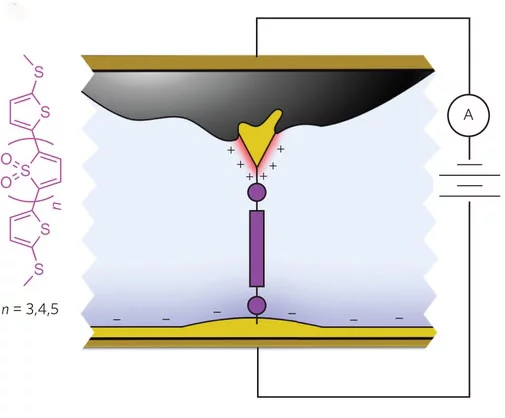
"Using a single symmetric molecule, anionic solution and two gold electrodes of dramatically different exposedsurface areas, we were able to create a diode that resulted in a rectificationratio, the ratio of forward to reverse current at fixed voltage, in excess of200, which is a record for single-molecule devices," said Jeffrey Neaton, directorof the Berkeley Lab’s Molecular Foundry and professor at the University ofCalifornia Berkeley. "The asymmetry necessary for diode behaviororiginates with the different exposed electrode areas and the ionic solution.This leads to different electrostatic environments surrounding the twoelectrodes and superlative single-molecule device behavior."
Firstmooted in 1974 by Mark Ratner and Arieh Aviram, an asymmetric molecule that couldact as a rectifier has been a long sought after goal, particularly as diodesform the basis of many microminiature electronic devices. Since then, a rangeof devices have been constructed, including single molecule diodes andtransistors. Operating at this nanoscale, though, such devices may emulatetheir macro counterparts, but that behavior is merely a simulation; at suchscales the electronic operation of these devices is governed more by quantuminfluences.
"Electron flow at molecular length-scales isdominated by quantum tunneling," said professor "The efficiency of the tunnelingprocess depends intimately on the degree of alignment of the molecule’sdiscrete energy levels with the electrode’s continuous spectrum. In a molecularrectifier, this alignment is enhanced for positive voltage, leading to anincrease in tunneling, and is reduced for negative voltage. At the MolecularFoundry we developed an approach to accurately compute energy-level alignmentand tunneling probability in single-molecule junctions. This method allowedmyself and Zhenfei Liu to understand the diode behavior quantitatively."

ZhenfeiLiu – a postdoctoral fellow at Berkeley Lab – and professor Neaton worked with LathaVenkataraman and Luis Campos from Columbia University to create theirhigh-performance rectifier diode using junctions prepared from symmetricmolecules attached to gold electrodes. To achieve the necessary asymmetricproperties required to operate as a diode, the researchers then altered thesurface area of the electrodes as they were exposed to an ionic solution. As aresult, a positive voltage increased the current significantly, whilst anegative voltage reduced current flow in an equally significant manner.
"Theionic solution, combined with the asymmetry in electrode areas, allows us tocontrol the junction’s electrostatic environment simply by changing the biaspolarity," said professor Neaton. "In addition to breaking symmetry, double layers formedby ionic solution also generate dipole differences at the two electrodes, whichis the underlying reason behind the asymmetric shift of molecular resonance.The Columbia group’s experiments showed that with the same molecule andelectrode setup, a non-ionic solution yields no rectification at all."
Thecombined Berkeley Lab-Columbia University research team is convinced that theway they have managed to produce a single-molecule diode sets the benchmark forfuture nonlinear nanoscale device tuning and development, with applicationsabove and beyond just junctions of single-molecule components.
"Weexpect the understanding gained from this work to be applicable to ionic liquidgating in other contexts, and mechanisms to be generalized to devicesfabricated from two-dimensional materials," said professor Neaton. "Beyond devices, thesetiny molecular circuits are petri dishes for revealing and designing new routesto charge and energy flow at the nanoscale. What is exciting to me about thisfield is its multidisciplinary nature – the need for both physics and chemistry– and the strong beneficial coupling between experiment and theory. With theincreasing level of experimental control at the single-molecule level, andimprovements in theoretical understanding and computational speed and accuracy,we’re just at the tip of the iceberg with what we can understand and control atthese small length scales."
The results of this research were recently published in the journal Nature Nanotechnology.
Source: Berkeley Lab





