With the rise of the lithium-based battery, demand for this soft, silvery-white metal – the lightest solid element in the periodic table – has exploded. With the race to zero carbon by 2050 gathering steam, forcing the electrification of transport, lithium will be an even more valuable asset in the next 30 years.
The supply of raw materials for batteries could even end up being a national security issue, too; China's global leadership on high-volume EV production has put it ahead of the game, and while the majority of ground-based lithium reserves are in the "lithium triangle" of Chile, Bolivia and Argentina, China controls more than half's the world's supply simply through investments and ownership. It has shown in the past that it's not afraid to wield commodity supplies as a weapon.
But as with other metals like uranium, land-based lithium reserves pale in comparison to what's out there in the sea. According to researchers at Saudi Arabia's King Abdullah University of Science and Technology (KAUST), there's about 5,000 times as much lithium in the oceans as there is in land deposits, and a newly developed technology could start extracting it cheaply enough to make the big time – while producing hydrogen gas, chlorine gas and desalinated water as a bonus.
The process relies on an electrochemical cell containing a ceramic membrane made from lithium lanthanum titanium oxide (LLTO), with pores just wide enough to let lithium ions through while blocking larger metal ions. “LLTO membranes have never been used to extract and concentrate lithium ions before,” says post-doctorate researcher Zhen Li, who developed the cell.
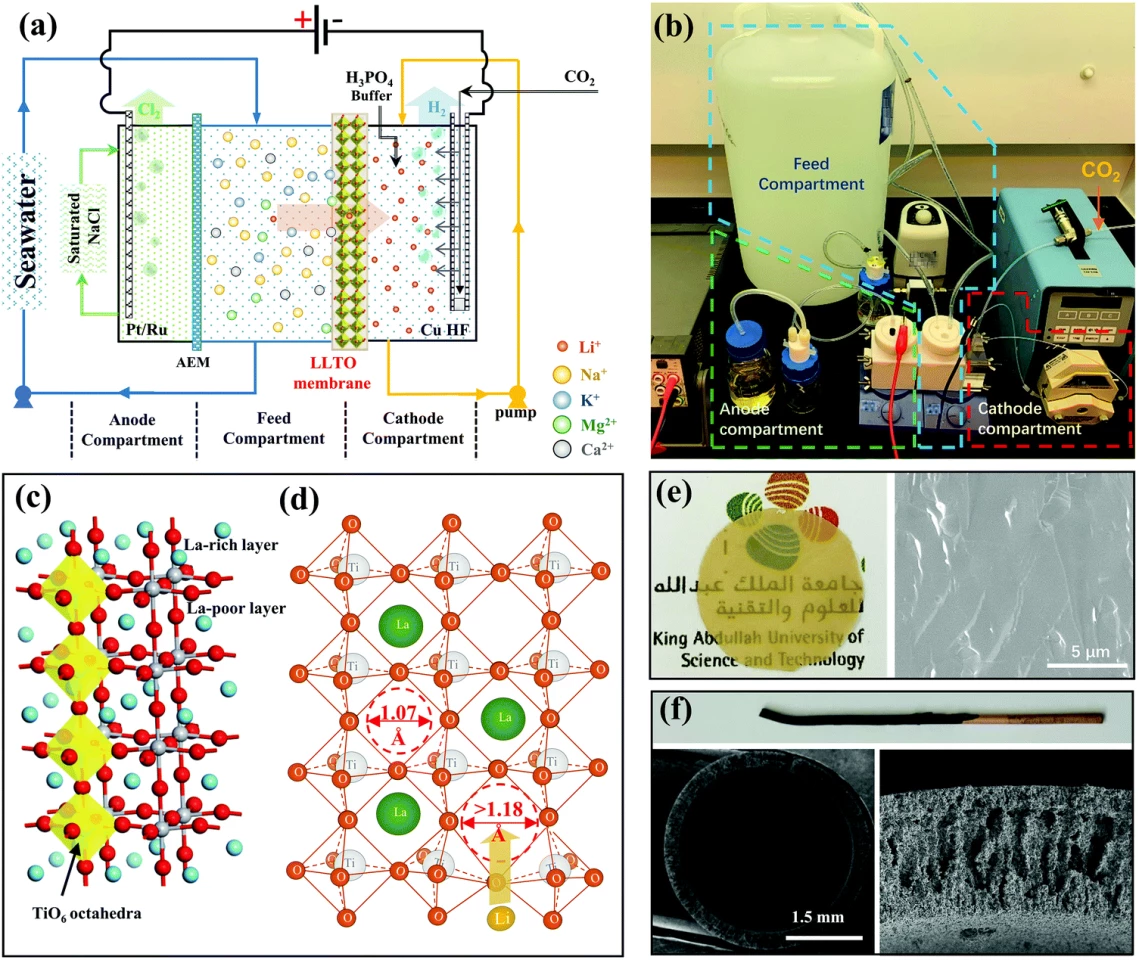
The cell has three compartments. Seawater flows into the first, from which positively charged lithium ions pass through the LLTO membrane into a second compartment holding a buffer solution and a copper cathode coated with platinum and ruthenium. Negatively charged ions, meanwhile, go through an anion exchange membrane into a third compartment containing a sodium chloride solution, where they're attracted to a platinum-ruthenium anode.
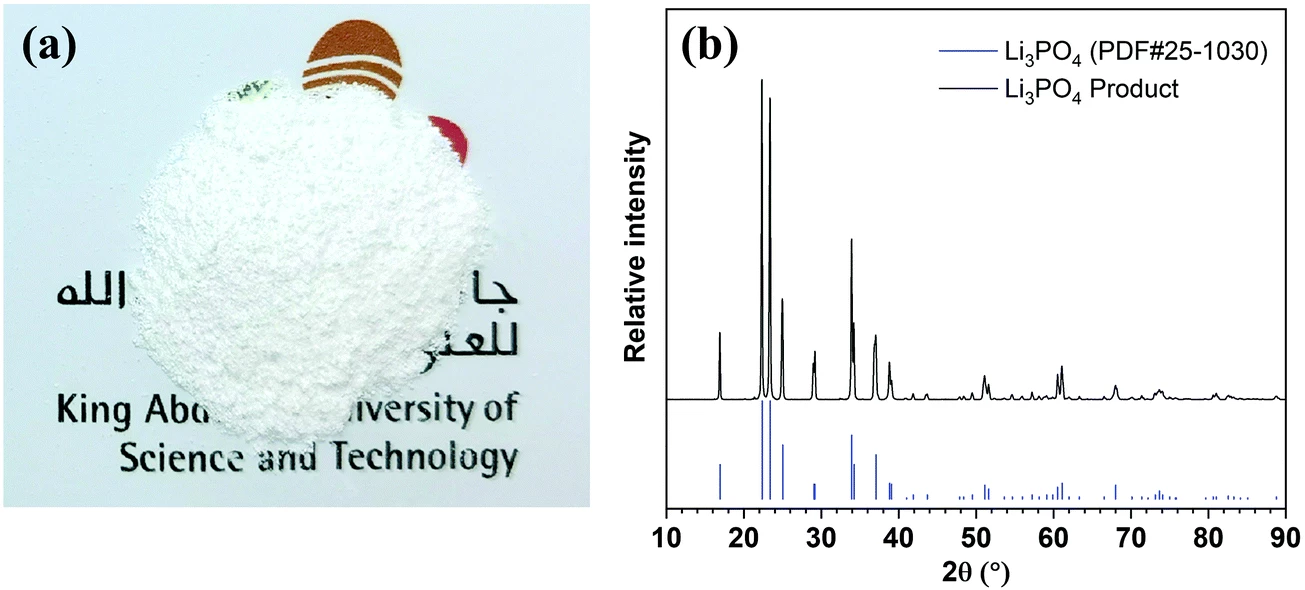
The lithium is pulled through the LLTO membrane toward the cathode when a current is applied, and the process generates hydrogen gas at the cathode and chlorine gas at the anode, both of which can be collected. Where lithium starts out at a concentration of just 0.2 parts per million in unprocessed seawater, experiments found that running it through this cell in five 20-hour stages enriched the concentration to over 9,000 parts per million, leaving a solution that was pH-adjusted, centrifuged, rinsed and dried to leave a lithium phosphate powder of 99.94 percent purity, meeting standards for battery-grade lithium phosphate.
According to the research team, the electricity required to produce a kilogram of lithium in this way (about 76.3 kWh) would cost around US$5 – and every kilogram of lithium would generate a bonus 0.87 kg of hydrogen gas and 31.12 kg of chlorine gas. At 2020 prices, these side products alone could sell for between US$6.90 and $11.70.
As for what that kilogram of lithium phosphate is worth, I couldn't find a price for 99.95 percent pure lithium phosphate, so take this with a grain of salt, but at 99.99 percent purity it's going for more than US$4,700 per kilo in small quantities. It'll be nothing near that in bulk wholesale – and indeed it seems most EV batteries use battery-grade (99.5 percent) lithium carbonate, which is more like US$14 per kilo. Make of that what you will.
Another side bonus is that the seawater that goes through just one stage of this process comes out with total salt concentrations under 500 parts per million. According to the researchers, this "implies that after lithium harvest, the remaining water can be treated as freshwater. Hence, the process also has a potential to integrate with seawater desalination to further enhance its economic viability."
How long will the gear last? Well, the researchers say they tested the LLTO membrane for more than 2,000 hours in Red Sea water and found "a negligible decay in performance," so things seem positive there. And will the equipment be expensive? It doesn't seem that way. "Although a rigorous economic analysis will be still necessary to include other capital and operating expenses," reads the paper, "it is arguable that the energy cost is the major expenditure in this process."
So, a machine that can pull battery-grade lithium out of the ocean for a negligible electricity cost that's more than offset by the hydrogen and chlorine gases that are produced in the process – and it pumps out desalinated freshwater too? Shut up and take my investment money, right?
Maybe. It should be noted that there are rare earth metals in the cell design itself. Also that the process of enriching seawater takes a hundred hours, and this device has only been tested on a lab bench at very small scale. But the researchers say there's plenty of room for optimization, and since the process appears to speed up considerably in the latter stages as lithium concentrations rise, it seems apparent that a richer feed content – the water from shale gas fields, for example – might tip the equation even further.
Will we end up regretting it if we pull all the lithium out of the oceans? Will all the fish go bipolar? Well, there's rather a lot of lithium in there. About 180 billion tons, which would supply the projected 2030 global demand for lithium more than 100,000 times over. So it'd certainly take a while – a while in which the bajillions of dollars currently being pumped into finding better battery solutions than lithium might well bear fruit.
Still, it certainly looks like a promising lithium production method with some very neat cherries on top.
The full paper is open access at the peer-reviewed journal Energy & Environmental Science.
Source: KAUST Discovery







