Graphene is already known for being incredibly strong, despite being just one atom thick. So how can it be made even stronger? By turning it into sheets of diamond, of course. Researchers in South Korea have now developed a new method for converting graphene into the thinnest diamond films, without having to use high pressure.
Graphene, graphite and diamond are all made of the same stuff – carbon – but the difference between these materials is how the carbon atoms are arranged and bonded together. Graphene is a sheet of carbon that’s just one atom thick, with strong bonds between them horizontally. Graphite is made up of graphene sheets stacked on top of each other, with strong bonds within each sheet but weak ones connecting different sheets. And in diamond, the carbon atoms are far more strongly linked in three dimensions, creating an incredibly hard material.
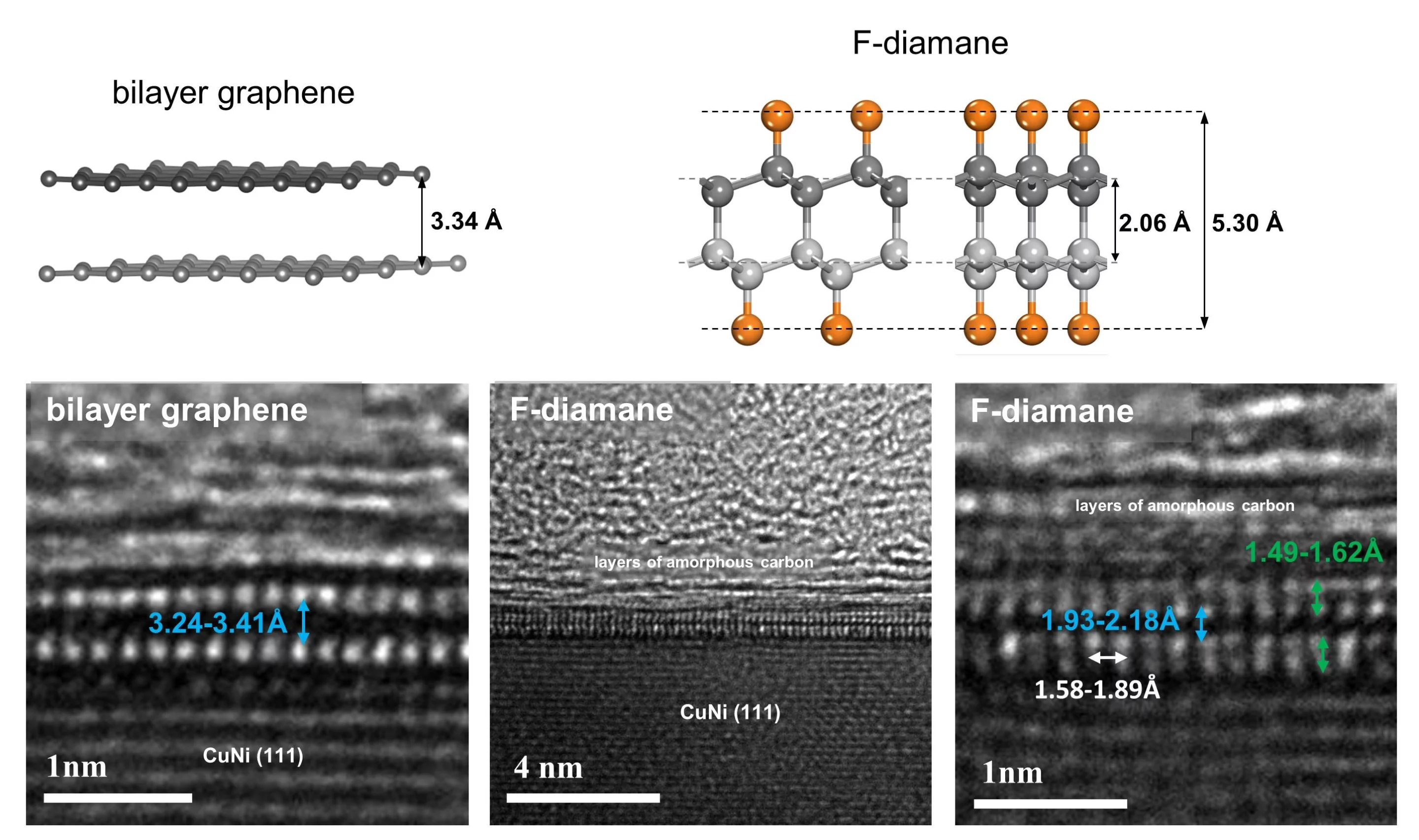
When the bonds between layers of graphene are strengthened, it can become a 2D form of diamond known as diamane. The problem is, this is normally not easy to do. One way requires extremely high pressures, and as soon as that pressure is removed the material reverts back into graphene. Other studies have added hydrogen atoms to the graphene, but that makes it difficult to control the bonds.
For the new study, researchers at the Institute for Basic Science (IBS) and the Ulsan National Institute of Science and Technology (UNIST) swapped out hydrogen for fluorine. The idea is that by exposing bilayer graphene to fluorine, it brings the two layers closer together, creating stronger bonds between them.
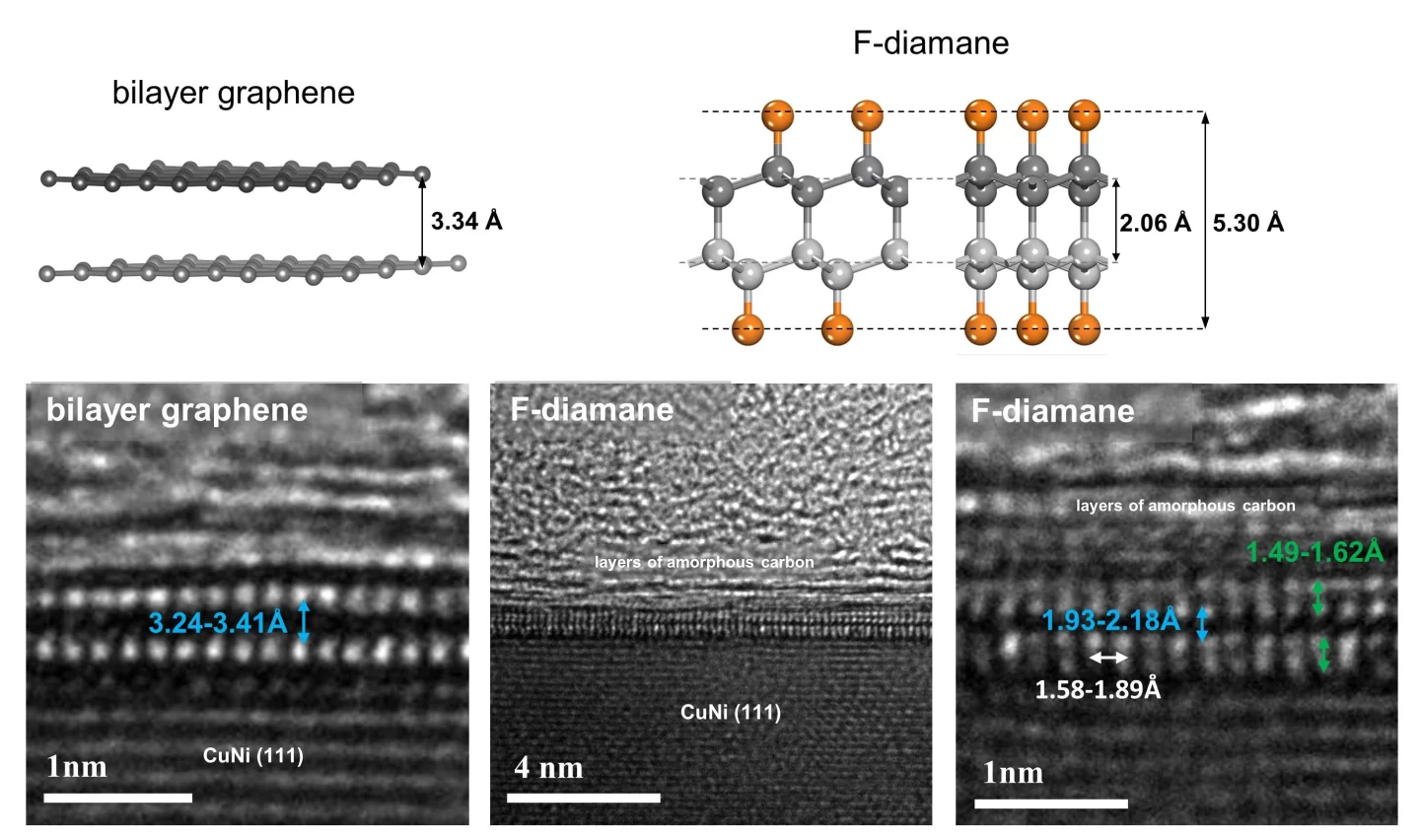
The team started by creating bilayer graphene using the tried-and-true method of chemical vapor deposition (CVD), on a substrate made of copper and nickel. Then, they exposed the graphene to vapors of xenon difluoride. The fluorine in that mixture sticks to the carbon atoms, strengthening bonds between graphene layers and creating an ultrathin layer of fluorinated diamond, known as F-diamane.
The new process is far simpler than others, which should make it relatively easy to scale up. Ultrathin sheets of diamond could make for stronger, smaller and more flexible electronic components, particularly as a wide-gap semi-conductor.
“This simple fluorination method works at near-room temperature and under low pressure without the use of plasma or any gas activation mechanisms, hence reduces the possibility of creating defects,” says Pavel V. Bakharev, first author of the study.
The research was published in the journal Nature Nanotechnology.
Source: IBS