Much mystery surrounds the biological processes that lead to hair loss in humans, but scientists continue to make discoveries that unravel these secrets and lay the foundation for technologies that may one day treat it. Researchers in Japan have shed yet more light on the topic, discovering a new mechanism by which dividing cells can drive hair follicles to exhaustion, subduing their regenerative abilities as we age.
The research was carried out by scientists at Tokyo Medical and Dental University and the University of Tokyo, who set out to explore the way hair follicle stem cells help us grow new hair, and the reasons why that important relationship can break down.
The sustainability of our hair follicle stem cells is reliant on a healthy combination of two types of cell division. In cyclic symmetric cell division, the hair follicle stem cells are split into two cell types with the same fate, while in asymmetric cell division, they are split into a differentiating cell along with a separate, self-renewing stem cell. These two cycles work in concert to keep the population of hair follicle stem cells alive and able to regenerate hair. By the same token, they can lead to their death when they don't work as they should.
“For proper tissue function, symmetric and asymmetric cell divisions have to be in balance,” says corresponding author of the study Emi Nishimura. “Once stem cells preferentially undergo one of either or, worse yet, deviate from the typical process of either type of cell division, the organ suffers. In this study, we wanted to understand how stem cell division plays into hair grows during aging.”
The scientists drew up experiments involving both young and aged mice, in which they monitored the hair follicle stem cells and tracked their division using two different types of assays. This revealed that while the young mice exhibited normal, healthy symmetric and asymmetric divisions, a strange, atypical type of cell division began to emerge in the older mice.
Digging further into the reasons behind this, the team investigated the role of a class of proteins called hemidesmosomes, which connect the cells to the surrounding extracellular matrix and help them sense their location in their environment. The team's analysis revealed that in the aging mice, these proteins became increasingly destabilized and drove the production of a different, aberrant cell type during the hair follicle stem cell division.
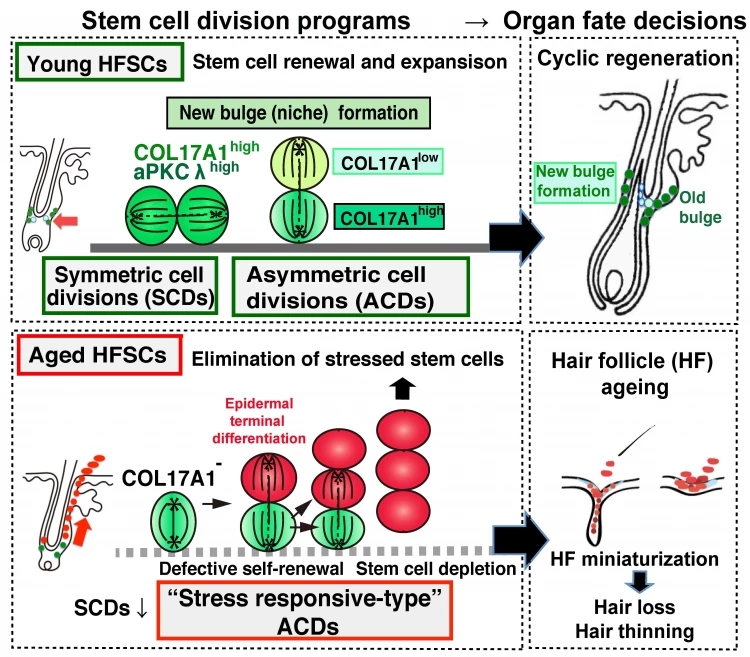
The upshot of this was that the hair follicle stem cells became exhausted as they aged, leading them to be cleared away from the body. In turn, this led to the thinning and loss of the hair follicles over time.
“These are striking results that show how hair follicles lose their ability to regenerate hair over time,” says first author of the study Hiroyuki Matsumura. “Our results may contribute to the development of new approaches to regulate organ aging and aging-associated diseases.”
The research was published in the journal Nature Aging.