While there are ways to alleviate some symptoms, there is currently no way to prevent or cure Parkinson’s disease, so the prospect of a one-off treatment that completely eliminates it is certainly an exciting one. While such a therapy remains a while off, scientists have demonstrated an exciting proof of concept in mice, whereby inhibiting a single gene as a one-time treatment eradicated the disease entirely, and kept it at bay for the remainder of their lives.
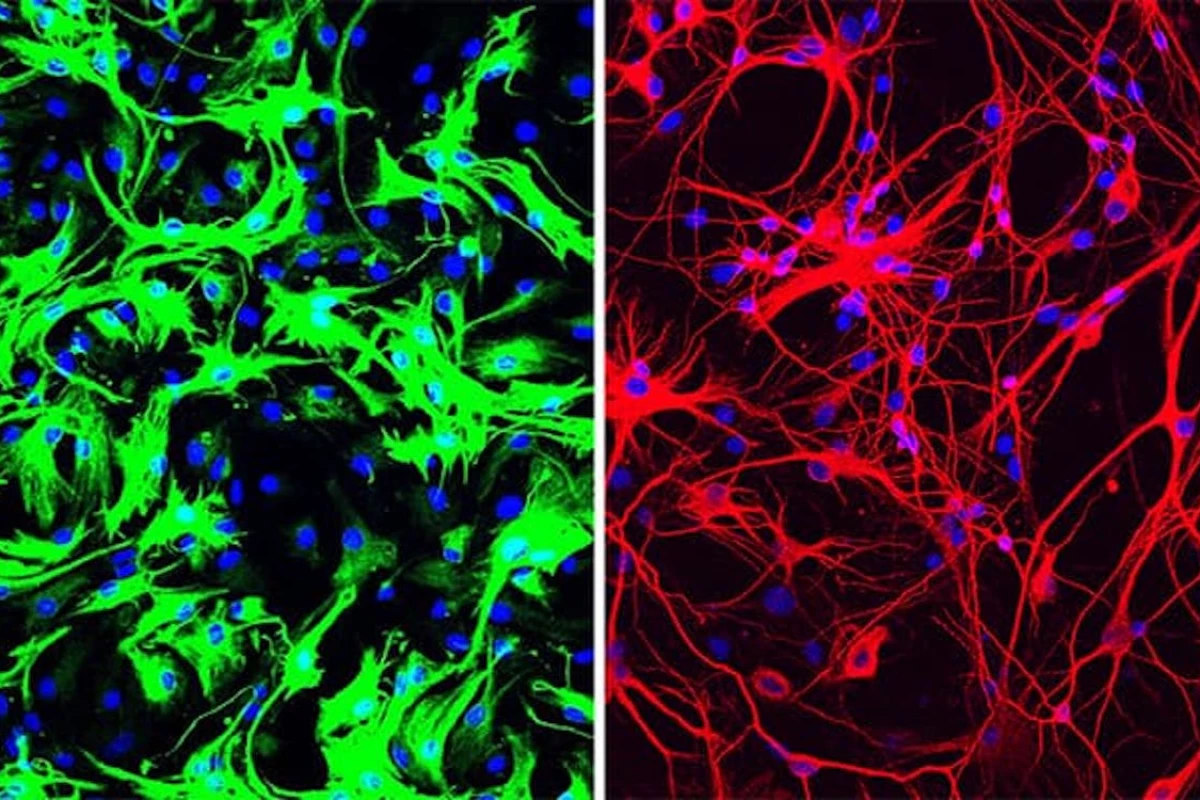
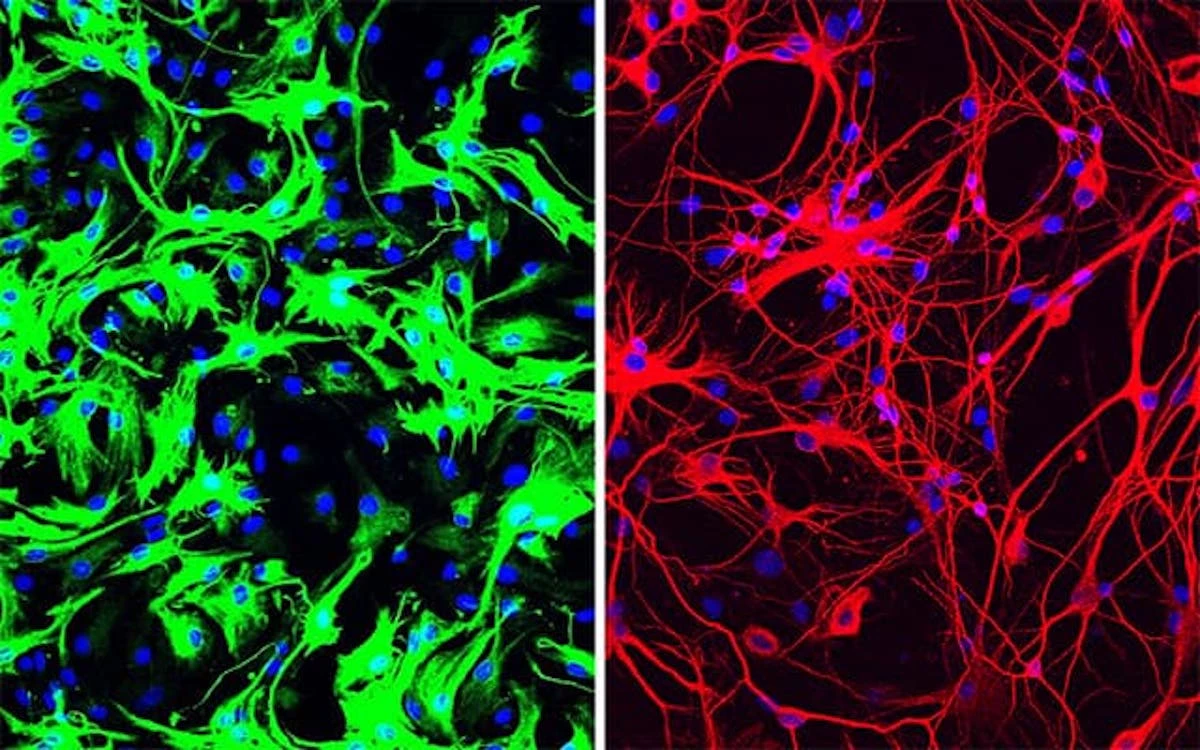
The research was carried out at the University of California, San Diego (UCSD), and centers on a protein called PTB, which plays a role in which genes are switch on and off in a cell. The team was experimenting with techniques whereby the gene that encodes for PTB is switched off so researchers can determine the flow-on effects of a reduction in the that protein on other cell types, and found peculiar results when working with connective tissue cells called fibroblasts.
In one experiment, the team created a cell line that was permanently lacking PTB, and after a couple of weeks found that there was only a small amount of fibroblasts remaining in the dish, which was brimming with neurons instead. Building on this, the team was able to use a single treatment to inhibit the activity of PTB in mice, which reprogrammed support cells in the brain called astrocytes into neurons that produce the neurotransmitter dopamine.
Parkinson’s is characterized by a declining ability of the brain to produce dopamine, which leads to the deterioration in motor skills often seen in sufferers of the disease. As such, a great deal of research focuses on arresting the slide, pursuing novel ways to shore up the supply of neurons that create dopamine, or protect the healthy dopamine-producing cells that remain.
In discovering a way to transform existing cells into much-needed neurons through a single treatment, the UCSD team has uncovered a particularly exciting possibility. The team used a non-infectious virus carrying artificial DNA to inhibit PTB is mouse models of Parkinson’s, targeting the mid-brain region that regulates motor control and reward behaviors, where a loss of dopamine-producing neurons usually takes hold.
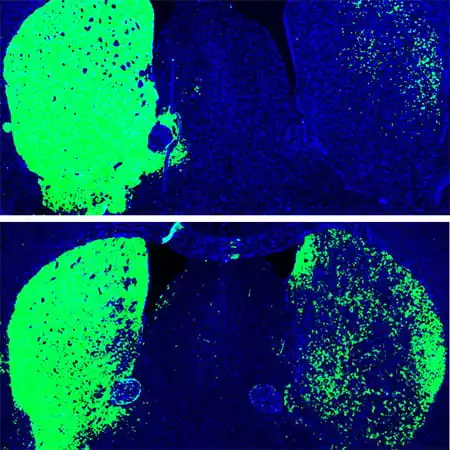
A small amount of astrocytes were transformed into neurons following this one-time treatment, which increased the total tally of neurons by around 30 percent. This had the effect of restoring dopamine to comparable levels to healthy mice, with the neurons continuing to grow and even trigger similar conversion processes elsewhere in the brain.
Follow-up observations to assess limb movement and response revealed that the mice had become free of Parkinson’s symptoms within three months, and remained that way for the rest of their lives. Meanwhile, a control group of mice that had been administered a mock treatment showed no improvement at all.
“I was stunned at what I saw,” says study co-author William Mobley from the UC San Diego School of Medicine. “This whole new strategy for treating neurodegeneration gives hope that it may be possible to help even those with advanced disease.”
The team notes that experiments in mice with Parkinson’s are very different to treating the disease in people, but now plans to continue improving the technique in mouse models with a view to exploring its potential in human trials.
“It’s my dream to see this through to clinical trials, to test this approach as a treatment for Parkinson’s disease, but also many other diseases where neurons are lost, such as Alzheimer’s and Huntington’s diseases and stroke,” says study author Xiang-Dong Fu. “And dreaming even bigger – what if we could target PTB to correct defects in other parts of the brain, to treat things like inherited brain defects? I intend to spend the rest of my career answering these questions.”
The research was published in the journal Nature.