Our deepening understanding of the different types of fat in the body continues to present new ways to potentially manipulate metabolic processes for better health outcomes, and new research on an existing asthma drug has opened up a promising new pathway. The authors have pinpointed a metabolite that turns a bad type of fat into a more favorable one, which raises hoped of new therapies for obesity with minimal side effects.
Carried out at Scripps Research, the new study centers on the difference between white fat and brown fat in the human body, and the metabolic processes that promotes one type over the other. White fat cells store excess energy in pot bellies and love handles, while brown fat cells burns away that stored energy to produce heat, helping address metabolic imbalances that can lead to conditions like obesity and diabetes.
We are seeing scientists make promising inroads into potential therapies that promote the formation of brown fat over white fat, or even convert one into the other like a “fat switch.” For the Scripps Research team, this pursuit led them to examine a catalog of existing drug compounds already tested for human safety. Advanced screening methods led them to one with some exciting potential, an FDA-approved asthma drug called zafirlukast.
Cell culture experiments showed that zafirlukast could turn precursor fat cells into predominantly brown fat cells, and also convert white fat cells into brown. The trouble was, however, that zafirlukast is toxic when administered in high doses, and questions remained over how exactly it was having these effects.
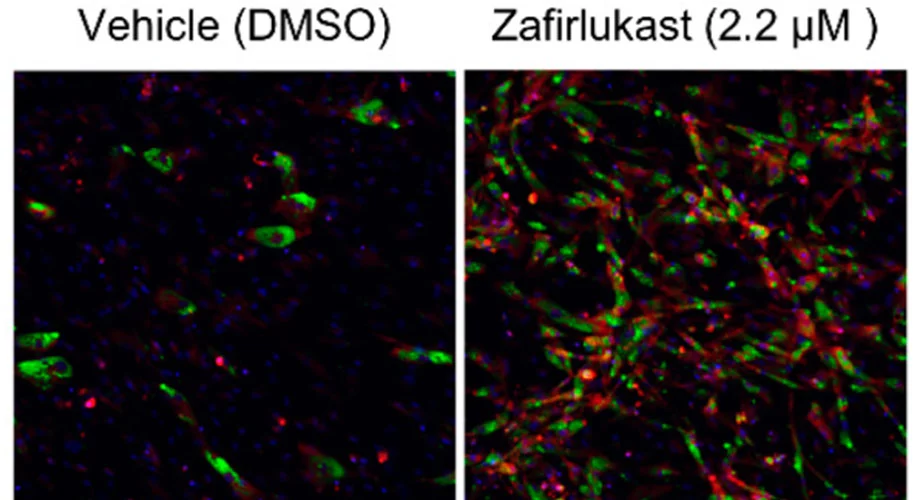
“We needed to use additional tools to break down the chemicals in zafirlukast’s mechanism,” says Kristen Johnson co-senior author of the study. “Framed another way, could we find a metabolite that was providing the same functional effect that zafirlukast was, but without the side effects?”
Another advanced screening method called drug-initiated activity metabolomics provided the scientists with some answers. It enabled them to sift through thousands of metabolites to zero in on one that triggered the formation of brown fat cells, and did so without causing any harm to the cell.
Called myristoylglycine, this metabolite was the only one of the thousands screened that had these particular characteristics. The metabolite is biosynthesized upon zafirlukast treatment, and mimics the fat-browning capabilities without the drug’s toxic effects. This bodes well for the researchers’ path toward therapeutic use, and also demonstrates how naturally occurring metabolites may be used to treat disease.
“The reason many types of molecules don’t go to market is because of toxicity,” says co-senior author Gary Siuzdak. “With our technology, we can pull out endogenous metabolites – meaning the ones that the body makes on its own – that can have the same impact as a drug with less side effects. The potential of this approach is even evidenced by the FDA’s recent approval of Relyvrio, the combination of two endogenous metabolites for the treatment of amyotrophic lateral sclerosis (ALS).”
The research was published in the journal Metabolites.
Source: Scripps Research




