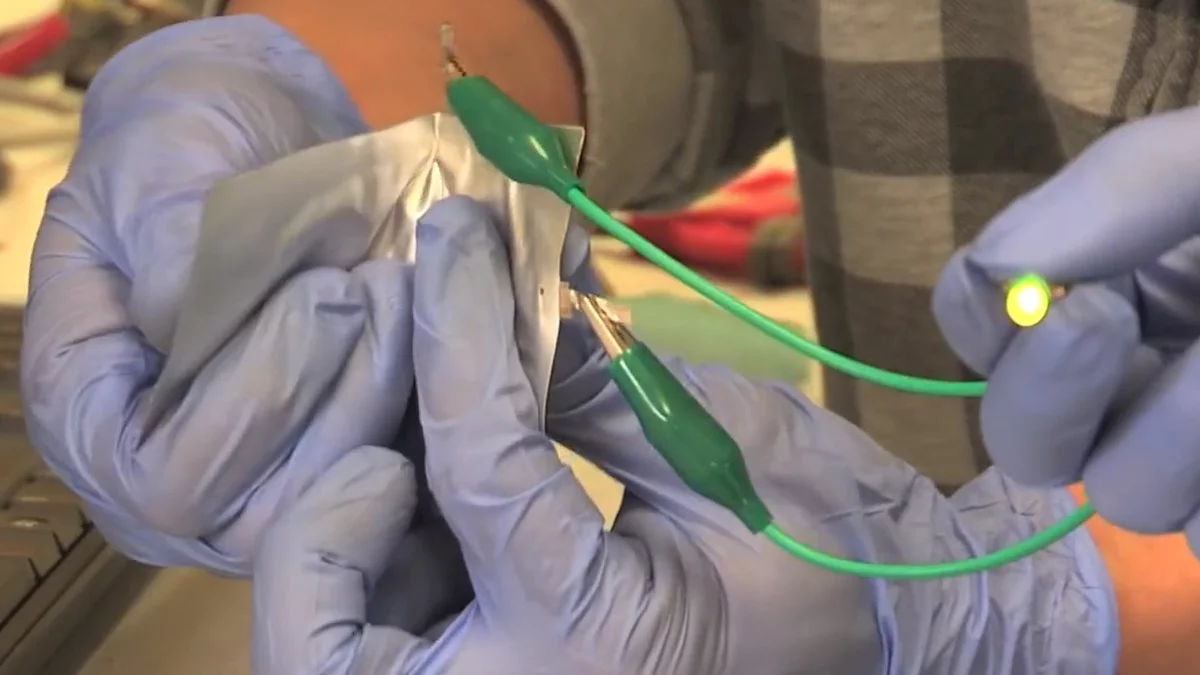
Researchers at Stanford University have created a fast-charging and long-lasting rechargeable battery that is inexpensive to produce, and which they claim could replace many of the lithium-ion and alkaline batteries powering our gadgets today. The prototype aluminum-ion battery is also safer, not bursting into flames as some of its lithium-ion brethren are wont to do.
The prototype battery features an anode made of aluminum, a cathode of graphite and an ionic liquid electrolyte, all packed within a flexible, polymer-coated pouch. And unlike lithium-ion batteries, which can short circuit and explode or catch fire when punctured, the aluminum-ion battery will actually continue working for a short while before not bursting into flames.
"The electrolyte is basically a salt that's liquid at room temperature, so it's very safe," said Stanford graduate student Ming Gong, co-lead author of the study.
Improved safety is great, but what many people want is a reduction in recharge times. The aluminum-ion battery hits the target here, too, with the Stanford team claiming "unprecedented charging times" of just one minute for recharging the prototype battery.
What about durability? The aluminum-ion battery has you covered there, too. Unlike typical lithium-ion batteries that last around 1,000 charge-discharge cycles, or other aluminum-ion battery lab attempts that usually died after just 100 cycles, the Stanford researchers claim their battery stood up to 7,500 cycles without a loss of capacity. This would make it attractive for storing renewable energy on the electrical grid.
"The grid needs a battery with a long cycle life that can rapidly store and release energy," team member Hongjie Dai explains. "Our latest unpublished data suggest that an aluminum battery can be recharged tens of thousands of times. It's hard to imagine building a huge lithium-ion battery for grid storage."
The experimental battery also has the added advantage of flexibility, which gives the technology the potential to find applications in the burgeoning field of flexible electronics.
Furthermore, the researchers point out that aluminum is a cheaper metal than lithium, and the aluminum-ion technology offers an environmentally friendly alternative to disposable AA and AAA alkaline batteries used to power millions of portable devices.
Currently, one of the prototype battery's biggest shortcomings is its voltage. Although Dai points out it is more than anyone else has achieved with aluminum, the battery only generates around two volts of electricity, which is around half that of a typical lithium-ion battery. However, the researchers are confident they can improve on this.
"Improving the cathode material could eventually increase the voltage and energy density," says Dai. "Otherwise, our battery has everything else you'd dream that a battery should have: inexpensive electrodes, good safety, high-speed charging, flexibility and long cycle life. I see this as a new battery in its early days. It's quite exciting."
The team's work is detailed in a paper published in the online edition of Nature and the battery can be seen in action in the video below.
Source: Stanford University