Last year MIT researchers reported improving the efficiency of lithium-air batteries through the use of electrodes with gold or platinum catalysts. MIT News is now reporting that in a continuation of that work, researchers have been able to further increase the energy storage capacity of lithium-air batteries for a given weight by creating carbon-fiber-based electrodes.
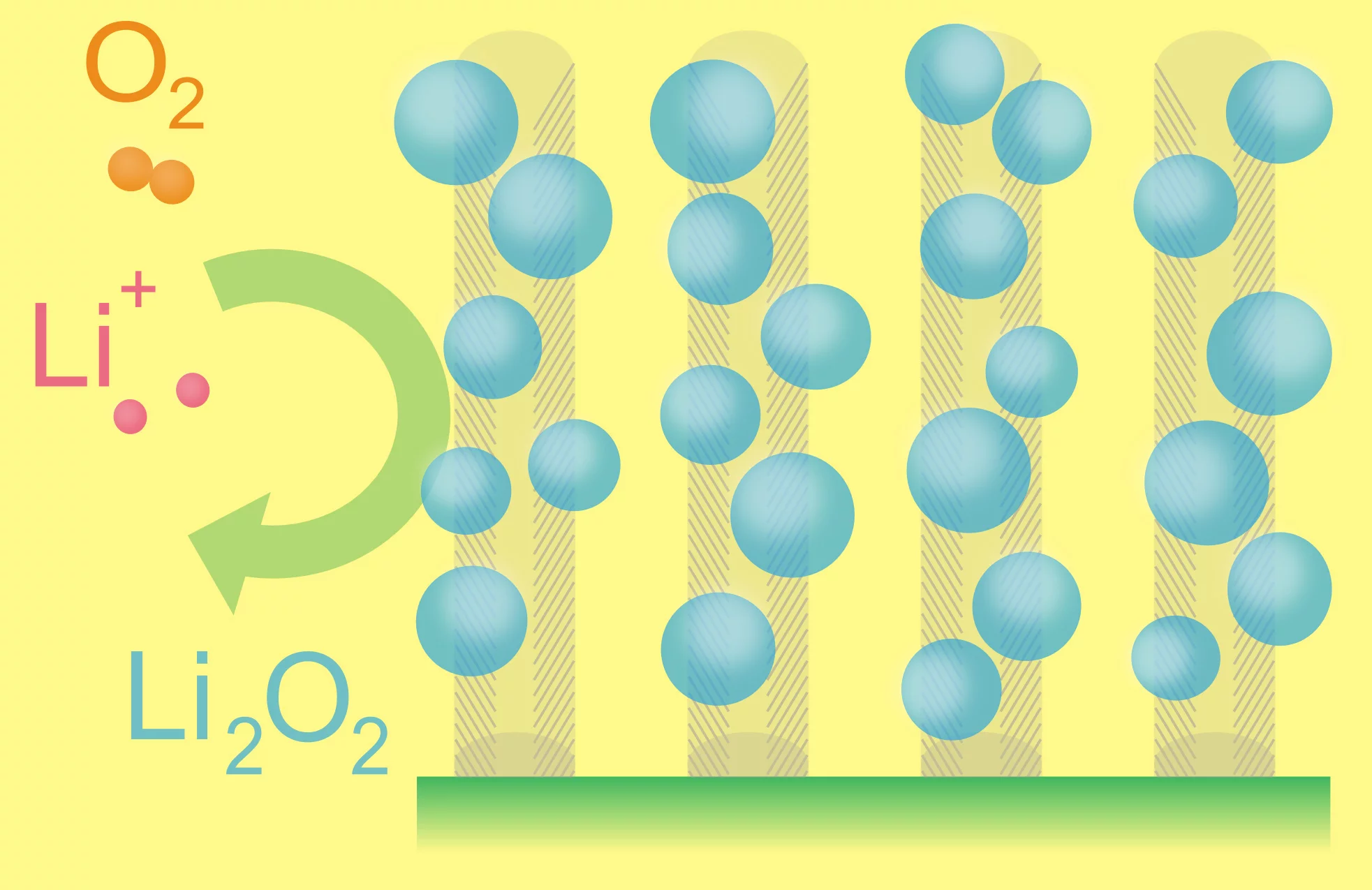
The superior energy density offered by lithium-air batteries is due to the coupling of a lithium anode to atmospheric oxygen through a porous carbon-based air cathode, instead of the heavy conventional compounds found in lithium-ion batteries. During battery discharge, lithium ions flow from the anode through an electrolyte and combine with oxygen at the cathode to form lithium oxides, which are inserted into the cathode. During recharging, the lithium oxides separate again into lithium and oxygen and the process can begin again.
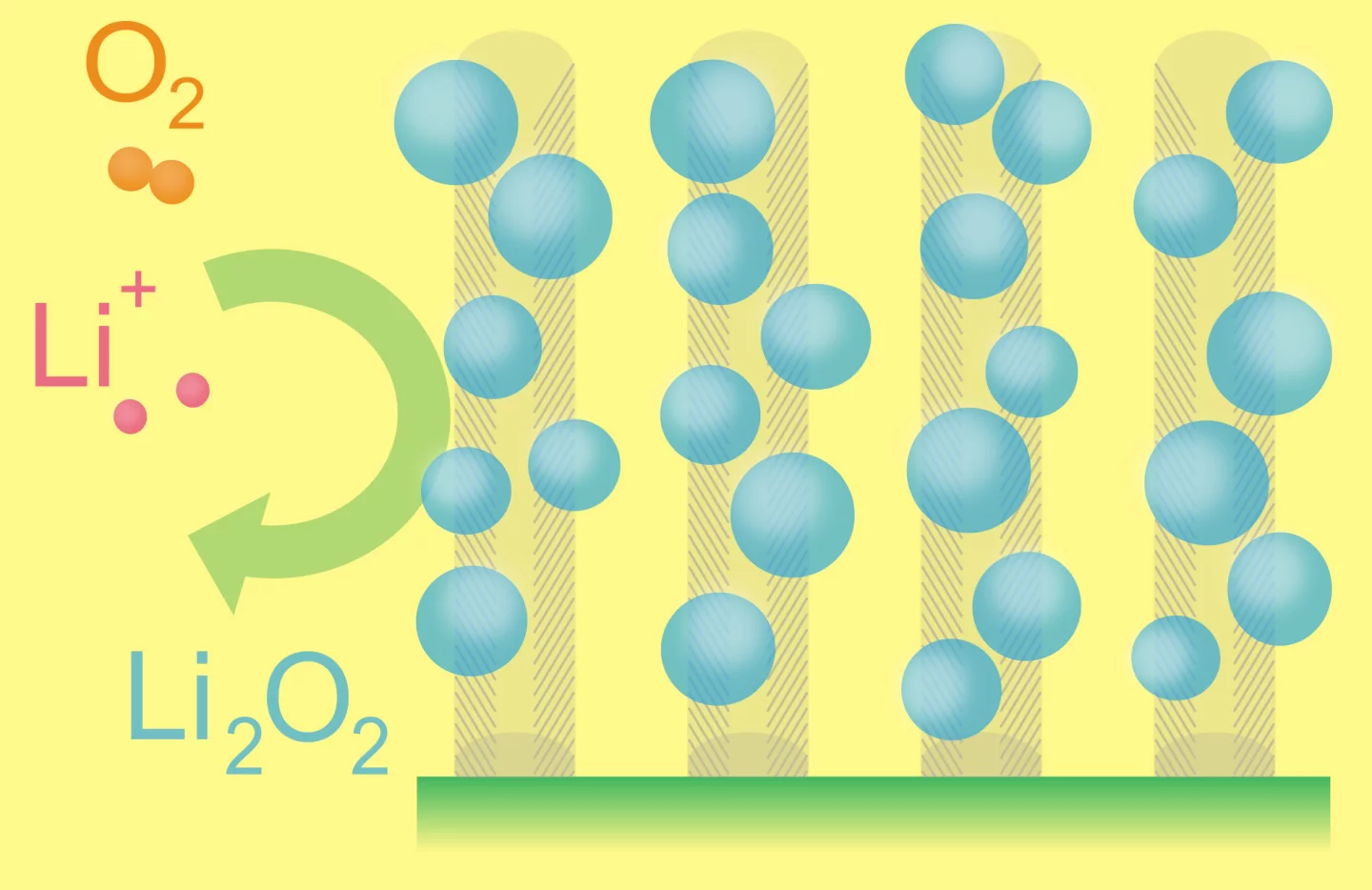
The carbon-based electrodes used in last year's research had only about 70 percent void space, but the new carbon-fiber-based electrodes are significantly more porous and boast more than 90 percent void space. This means the carbon-fiber-based electrodes can more efficiently store the lithium oxide that fills the pores as the battery discharges.
"We grow vertically aligned arrays of carbon nanofibers using a chemical vapor deposition process. These carpet-like arrays provide a highly conductive, low-density scaffold for energy storage," Robert Mitchell, a graduate student in MIT's Department of Materials Science and Engineering (DMSE), told MIT News.
The researchers claim the carbon-fiber-based electrode can store four times as much energy for its weight compared to current lithium-ion battery electrodes. However, Yang Shao-Horn, the Gail E. Kendall Professor of Mechanical Engineering and Materials Science and Engineering and senior author of the paper says further work still needs to be done before the advances make it from the lab and into a practical commercial product.
A paper describing the new findings appears in the journal Energy and Environmental Science.
Source: MIT News