Engineers at MIT have developed a device that generates electricity using a completely new mechanism. “Particles” made of carbon nanotubes are dunked in an organic solvent, which induces a current to potentially power small robots or drive chemical reactions.
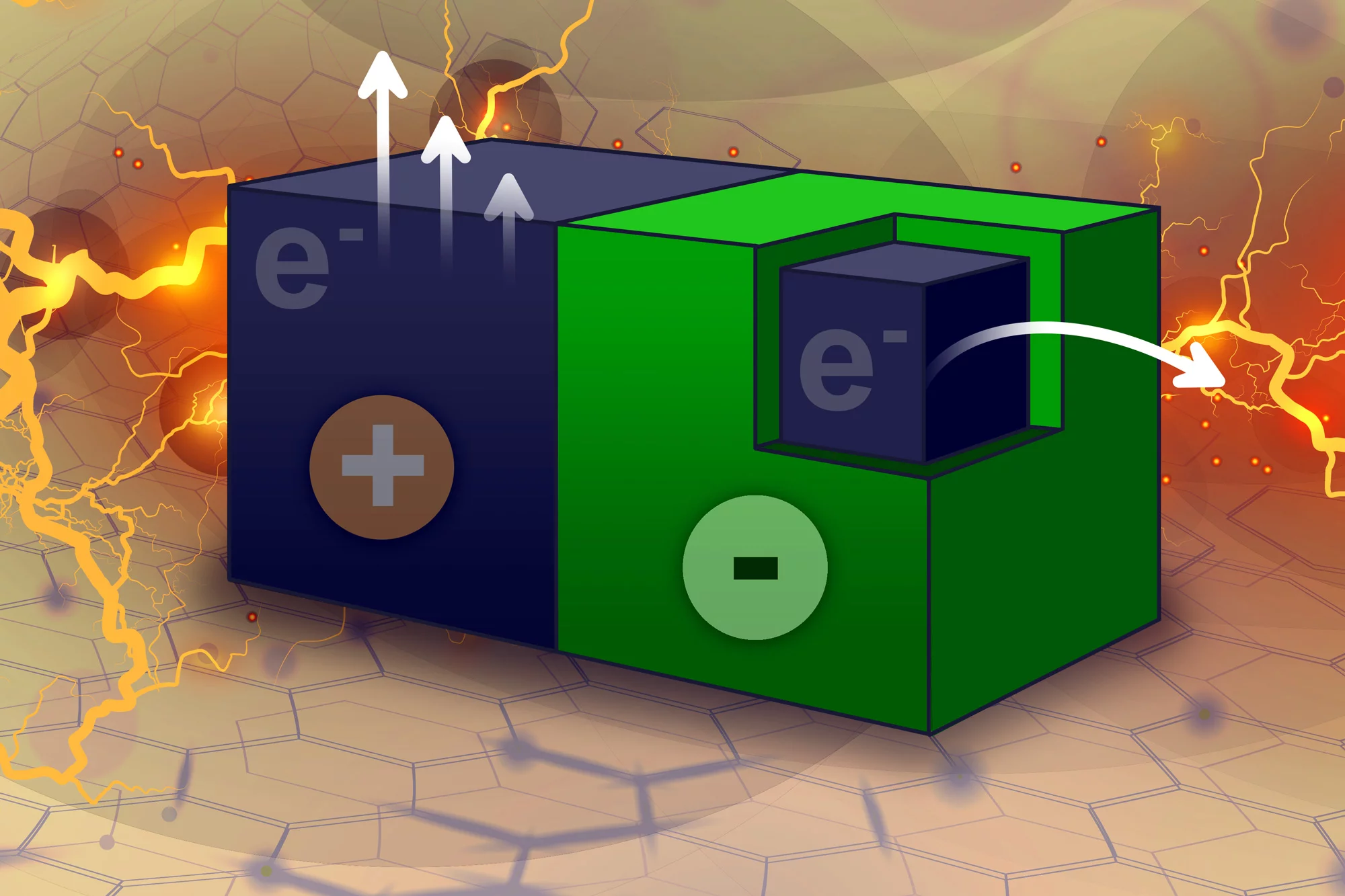
Carbon nanotubes are known to be great conductors of electricity, and the MIT researchers found a way to tap into that. The team coated half of the nanotube material in a Teflon-like polymer, which allows electrons to flow from the coated half to the uncoated half. When this material is immersed in an organic solvent, the liquid begins pulling electrons out of the uncoated part, and in response electrons are transported from the coated section – thereby generating a current.
“The solvent takes electrons away, and the system tries to equilibrate by moving electrons,” says Michael Strano, senior author of the study. “There’s no sophisticated battery chemistry inside. It’s just a particle and you put it into solvent and it starts generating an electric field.”
With the mechanism identified, the team then sought to create a device that makes use of it. They made a sheet of carbon nanotubes, coated one surface with the polymer, then cut the sheet into small particles 250 micrometers wide, creating two-faced objects sometimes called Janus particles.
These particles were then placed in a test tube of the solvent, where they form arrays by the hundreds to create a “packed bed” reactor. In tests, the team found that the system can currently generate around 0.7 volts per particle, which was enough to power an alcohol oxidation reaction. This reaction, where alcohol is converted into an aldehyde or a ketone, is common in the chemical industry.
“Because the packed bed reactor is compact, it has more flexibility in terms of applications than a large electrochemical reactor,” says Ge Zhang, an author of the study. “The particles can be made very small, and they don’t require any external wires in order to drive the electrochemical reaction.”
The team says that the system could eventually be put to use powering small robots that scavenge energy from their environment.
The research was published in the journal Nature Communications.
Source: MIT