The human contains different types of fat cells, and some are more desirable than others. White fat is the type that builds beer bellies and love handles, while the more beneficial brown fat is one that burns more easily to generate body heat. Scientists have developed a new gene-editing technique that converts a subject's own white fat cells into brown fat cells, which could then be transplanted as a way of treating obesity and diabetes.
Methods of transforming white fat into brown fat hold huge potential when it comes to obesity treatments, and we’ve seen quite a few promising advances in this area. These have involved hormones that mimic the effects of exercise, injections of nanoparticles directly into fat deposits and gene therapies that act like a fat switch.
One of the more interesting examples was a 2018 study, in which a team at Columbia University showed how white fat could be removed from the body and cultured in media containing growth factors to turn it into brown fat instead. The new research from scientists at the Joslin Diabetes Center mirrors this approach, but uses a variant of the gene-editing tool CRISPR to intervene with the cells still in their early developmental stage.
The white fat cells were harvested while in their progenitor phase, meaning they hadn’t yet matured into their final form. The gene-editing system was used to increase the expression of UCP1, a gene that causes white fat cell progenitors to convert into brown-fat-like cells. The team calls the result of this process HUMBLE cells (human brown-like) fat cells.
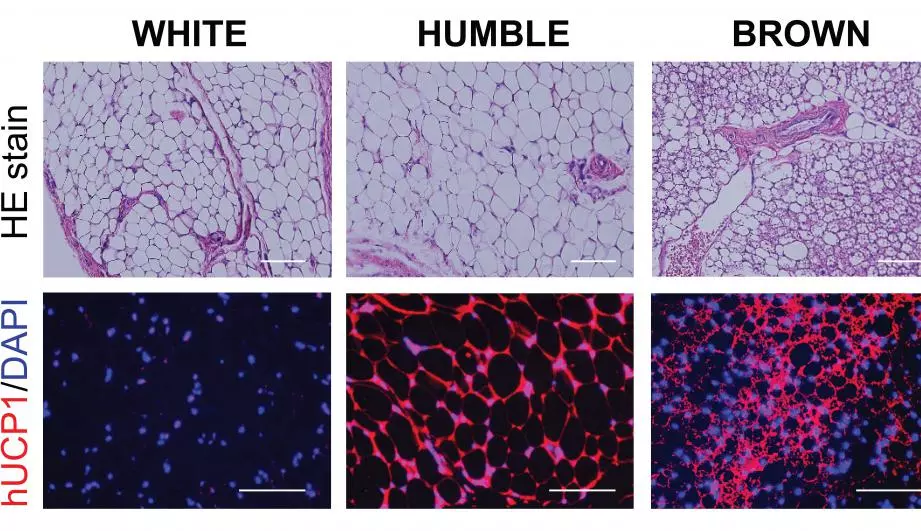
These were then transplanted into mice to see how they fared, with the team finding that they functioned in much the same way as the rodent’s existing brown fat cells. The scientists then ran another experiment where they transplanted the HUMBLE cells into one group of mice, and white fat cells into another group, with both placed on a high-fat diet.
The HUMBLE recipients put on less weight than the mice with transplanted white fat cells, and were comparable to another group that received transplanted brown cells. Interestingly, the team was able to show that these positive results were for the most part because of the signals being sent from the transplanted cells to the existing brown fat cells.
"Cells in different tissues communicate with each other," says senior author of the study Yu-Hua Tseng. "In this case, we found that our transplanted HUMBLE cells secrete a molecule called nitric oxide, which is carried by red blood cells to the endogenous brown cells and activates those cells."
While this technique is a promising approach to treating obesity, it also has potential to treat type 2 diabetes, of which obesity is the main cause. The team also found that the mice given the HUMBLE cells in the experiments exhibited greater sensitivity to insulin and an increased ability to clear glucose from the blood, both of which are hindered in sufferers of type 2 diabetes.
The team sees a couple of pathways toward clinical use for this new therapy. One of the avenues being explored involves encapsulating generalized cells into biomaterials that prevent rejection by the host’s immune system, as a more cost effective approach than tailoring the treatment to each individual. The other is to possibly have existing white fat progenitor cells directly express the UCP1 gene through gene therapy, so those cells take on the properties of the HUMBLE cells.
"Employing cell-based or gene therapies to treat obesity or type 2 diabetes used to be science fiction," says Tseng. "Now scientific advances, such as CRISPR gene-editing technologies, will help us to improve the metabolism, the body weight, the quality of life and the overall health of people with obesity and diabetes."
The research was published in the journal Science Translational Medicine, while you can hear Tseng discuss the study in the video below.
Source: Joslin Diabetes Center




