Stem cells that can be engineered to take on different properties continue to show huge potential in all areas of medicine, ranging from Parkinson's disease treatments to regenerative therapies for injured tendons. Scientists adapting this technology for difficult-to-treat tumors have developed a highly efficient off-the-shelf approach that showed "profound efficacy" in mouse models of aggressive brain cancer, laying the groundwork for clinical trials.
The idea of using stem cells engineered to target tumor cells is starting to gain traction as a way of tackling difficult-to-treat cancers, and highly aggressive brain cancers known as glioblastomas (GBMs) are a prime candidate. Many drugs are unable to access these tumors due to the blood-brain barrier that prevents their passage, and while surgery can be effective at eliminating the initial tumor the rate of tumor recurrence is more than 90 percent.
The authors of the new study were exploring ways of intervening post-surgery to lower this rate of recurrence, leading them to the idea of therapies involving engineered cells. Many of these approaches involve harvesting the patient's own cells and exposing them to reprogramming factors that supercharge their cancer-fighting abilities before returning them to the body, with engineered CAR T cell therapy among the most common and promising approaches.
The trouble is that glioblastoma and other aggressive cancers can advance very rapidly, offering a very short window for these very complicated types of therapies to be carried out. A more effective approach might be to have a bank of cells sourced from healthy donors that are engineered to tackle different cancers, and are ready to be deployed soon after a patient's diagnosis. This is the vision being pursued by the authors of the new study, from Brigham and Women’s Hospital and Harvard Medical School.
First, the scientists identified special receptors on cancer cells circulating through the bloodstream they call "death receptors." They then took stem cells from healthy donors and engineered them to release proteins that bind to these receptors to trigger the death of the cancer cells. They also incorporated a safety switch function into the stem cells that causes them to self-destruct once their work is done, while further driving cancer cell death.
These cells were packaged into biodegradable hydrogel capsules and administered to mice with glioblastoma after surgical removal, with all that received the treatment still alive 90 days later. A control group of mice that underwent surgery but didn't receive the stem cell treatment had a mean survival time of 55 days. The scientists also treated the mice with varying doses of the stem cells to ascertain the safety of the therapy, and detected no signs of toxicity.
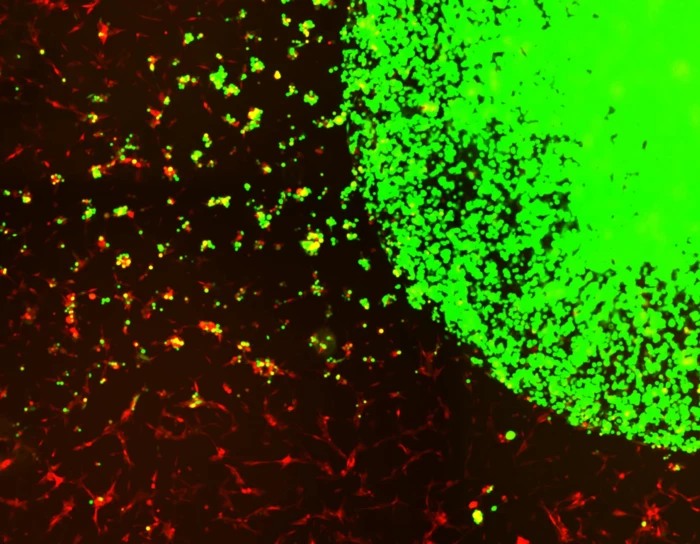
“This is the first study to our knowledge that identifies target receptors on tumor cells prior to initiating therapy, and using biodegradable, gel-encapsulated, ‘off-the-shelf’ engineered stem cell based therapy after GBM tumor surgery,” said study author Khalid Shah. “In the future, we will be applying this strategy to promptly identify target receptors after one receives a GBM diagnosis, then administer a gel-encapsulated, off-the-shelf, engineered stem cell therapeutic from a pre-made reservoir.”
The treatment showed "profound therapeutic efficacy," the researchers write in their paper, saying the results pave the way for clinical trials in humans within the next two years. They hope that the research leads to not just effective new treatments for glioblastoma, but a variety of cancers that are able to overpower current therapies.
“Beyond this therapy’s significant exhibited success rate, these findings suggest that we can use stem cells from healthy individuals to treat cancer patients,” said Shah. “This work lays down a foundation to begin building an engineered therapeutic stem cell biobank targeting different receptors on tumor cells and the immune cells in the tumor microenvironment that we will one day be able to use to treat a wide range of difficult-to-treat cancers like GBM.”
The research was published in the journal Nature Communications.
Source: Brigham and Women’s Hospital