The ongoing organ shortage crisis has fueled a lucrative black market that generates over US$1 billion in profits every year. 3D-printed organs, despite their promise, remain a lab experiment for now, and while scientists have been cultivating cells in Petri dishes, they have yet to figure out how to replicate nature's cocktail of life-giving signals. Enter a new medical breakthrough that could one day solve this problem by incubating human organs in pigs – but should we let animals pick up the slack for us in the first place?
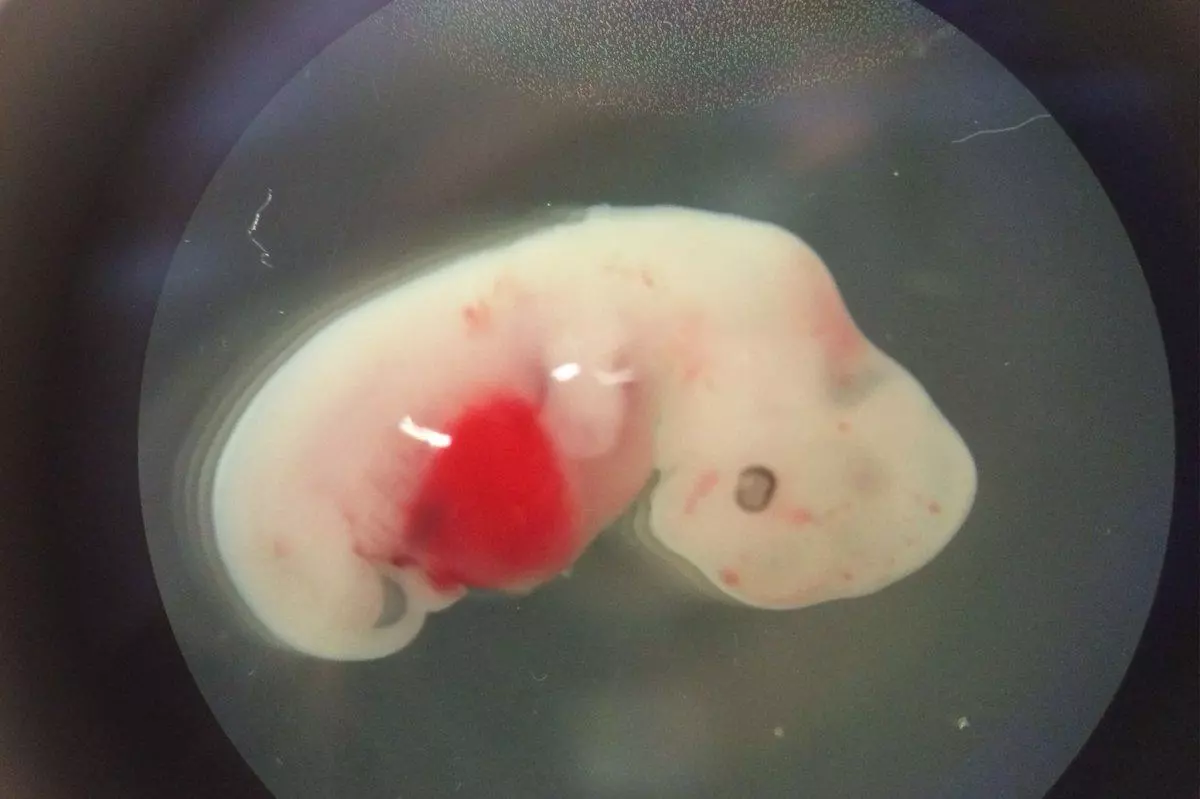
A human-pig chimera created by Salk Institute researchers marks the first time that human stem cells have been able to grow and differentiate in a large animal.
"Prior to our study, we were really not sure whether the human cell had the ability [to grow and differentiate] entering into the pig development, because pigs and humans are very distant species — they diverged about 95 million years ago," lead author Jun Wu told Australia's ABC news. "Before we dream about human organs, we need to have human cells that can contribute and differentiate inside the pig embryos."
The researchers chose the pig because its organs are similar in size to ours and it reproduces quickly and in large numbers, which could theoretically see organs being grown in less than four months assuming scientists can figure out how to get around the difference in gestation periods – these last 114 days and nine months for pigs and humans respectively. In addition, the animal has also played an important role in the development of human medicine, with their valves, for example, used in heart transplants.
To create the chimera, the researchers injected human stem cells into pig embryos, which were then incubated in surrogate sows for up to four weeks before being terminated.
While the study was a breakthrough in itself, the researchers stress that they are a long way from creating a walking breathing human-pig. For starters, the chimeras contained more pig than homo sapien. Wu estimates that there was an estimated one human cell for every 100,000 pig cells, a ratio too low for use in any practical application. Plus, it took much longer than anticipated to create the successful chimeras. The researchers had expected the process to take a year, but in reality it took them four years and more than 1,500 pig embryos. A huge part of this had to do with getting the timing right to ensure the survival of the human stem cells in the pig embryos. So rein in your imagination – the chimeras of Dr Moreau aren't coming to a lab near you anytime soon.
"I think we are far away from that because until say a few months, there was not any publication that could show that a human cell could grow inside the blastocyst of another animal," says lead researcher and gene expressions expert Juan Carlos Izpisua Belmonte in an interview with Scientific American. "It was just in the last few months that our group and another group has been able now to show the first examples that human cells can colonize the early embryo of another species."
Unlocking the origins of life
While the genetic distance between pigs and humans might be the cause of the low human-cell count, the researchers believe gene-editing technology such as CRISPR could help bridge this gap, as demonstrated by the earlier rat-mouse hybrids they created with the technology. First, mouse embryos were edited to turn off the genes that make the heart, eyes and pancreas, after which rat pluripotent stem cells (i.e. master cells that can potentially produce any cell or tissue needed for body repair) containing the missing genes were inserted into them. The resulting chimeras were born with rat cells in the aforementioned organs, giving the researchers reason to believe that growing human organs for transplant in animals could succeed under the right conditions.

What was even more interesting was the fact that the rat pluripotent stem cells generated gallbladders in a mouse, an organ that has been absent in rats for the past 18 million years. One explanation for this is that somewhere along the course of its evolutionary journey, the organ was no longer considered necessary and thus the mechanism responsible for its development was suppressed, only to be triggered when it was placed in a different biological environment.
And there's no reason this couldn't happen in human beings either. "We generate organs and tissues now that we see as human, but maybe we have the ability to do something more," Wu says in an interview with Wired. "Those abilities that have been suppressed during evolution because we don't need them anymore could be unlocked. You just need a different environment."
Apart from addressing the organ transplant shortage, the findings could also offer researchers a new way to study the early-stage development of human cells and organs, as well as a novel way of testing experimental drugs. However this remains a "what if" scenario unless the embryos are allowed to complete gestating. For this to happen, the US government will need to review its position on chimeric research. As of 2015, the US National Institutes of Health, has halted funding on chimera research involving the introduction of human pluripotent cells into animal embryos, which explains why the SALK Institute study was funded by private sources instead.
Looking beyond numbers
The organ shortage crisis is a complex problem that goes beyond the lack of available donor organs. There are many unseen factors that determine whether or not a patient on a waiting list gets a transplant. For example, not every donated organ finds its way to a patient. Case in point, a recent study by a group of transplant surgeons and medical researchers in Massachusetts revealed that not only were hospitals across the US throwing away imperfect – albeit useable – organs, they were also dropping the extremely ill from their waitlists, the reason being that the sicker the patient, the riskier the surgery. Poor transplant outcomes not only hurt reputations but also jeopardize Medicare ratings and funding – an important source of income for hospitals, as documented by STAT.
While the federal agency responsible for the ratings system has since revised its benchmark following the release of the paper, the fact remains that getting a transplant is often subject to factors beyond a patient's control, a problem that affects not just US citizens. In Canada, for instance, the lack of a national centralized waiting list means that geography is prioritized over need; how long one has to wait for an organ depends largely on where a patient lives, and not the severity of their condition. The ability to produce custom organs on demand could slash wait times dramatically for many of these patients.
The solution that the SALK Institute study offers remains a hypothetical one for now, but that hasn't stopped researchers from raising animal welfare and ethical concerns, which include the worry that the introduction of human cells could lead to the creation of animals who can think like humans, or that the animals might pass human genes along to their offspring, as one scientist noted in an interview with CNN. But that's just one side of the debate.
In the event that science advances to the point where fault-free organs can be grown in animals and made readily available to those who need them, should society deny patients this option based on bioethical grounds? Since we've already heard from the scientists, I decided to ask patients with liver disease what they thought of this breakthrough and whether they'd go for it. Here's what some of them said:
Bob, a 67-year-old who's undergone two liver transplant operations, said he's against it on the grounds that more should be done to encourage people to donate their organs instead:
"No! There are more than enough organs in this country. Make donating mandatory like having a drivers license to drive or paying taxes. Create an opt out if you have contrary beliefs, which means you also won't be receiving an organ during your lifetime. Unless, of course, your life depends on a transplant then all of a sudden your beliefs change!"
Others, like Olivia, were on the fence:
"There's a real ethical problem about what would be done to the animal in order for him to be able to produce an organ that would be compatible with a human. I assume the animal would be killed in order to provide their organ to a human and I would be against animal farms that existed for the sole purpose of harvesting animal organs. I'm against animal experimentation in general though.
However, I think that it's a moot question because in a few years we will be able to figure out how to hook up 3D-printed organs, Plus, research looks good for developing meds that will heal damaged organs.
You can't require people to donate their organs for many reasons. Religious and spiritual reasons at the top. With education we should be able to convince more people to give up their organs at death but I have a right to say no, I do not want to donate my organs just because I do not want to. It's my body, not the public's or the government's. And no, I do not want any organs given to me.
But even if a person didn't want to donate their organs, they should still be allowed to receive them."
And then there were those who were in favor of the life-saving opportunities such a breakthrough offered:
"There are NOT enough livers today. In 2016, 1,212 liver patients died waiting for a liver and 1,433 got too sick to qualify for a liver transplant. 1/3 of us did NOT make it. And then there's the lucky 7,841 who actually did get transplanted in 2016.
Donate Life has made great strides to increasing the Donor Registry to 52% of all US adults, but the fact remains only 1-2% qualify to become organ donors, i.e., on life support in the hospital.
I am so grateful to modern science which extended the life of my husband at 46 years old. Organ donation not only saved my life, but that of my family. Our kids were 11, 12 and 13 then. We were the lucky 2/3. These 4.5 years post-transplant have been absolutely glorious!
The way I look at it is God gave us the brains to develop technology like organ transplantation and now xenotransplantation. Would I grab a pig liver if it meant saving my life and keeping our family intact? You betcha!"
Wu and Izpisua Belmonte talk about their work in the video below.
The study was published in Cell.
Source: SALK Institute