Flow batteries have the potential to help store energy on a large scale, and might be particularly useful to back up renewable energy sources, but there are a few issues still to overcome. Engineers at Stanford have developed a new type of flow battery that might be scalable, safe, efficient and inexpensive, using a metal mixture that remains liquid at room temperature.
In a flow battery, the cathode and anode are in fluid form and are kept in external tanks, to be pumped into the main cell of the battery when needed. There, the two liquids are separated by a membrane that selectively allows them to exchange electrons to either charge or discharge the energy.
These devices may be able to store huge amounts of energy in future, but the chemicals used are often toxic, expensive, and difficult to handle. The Stanford team designed the new flow battery to overcome those problems, using a unique combination of materials.
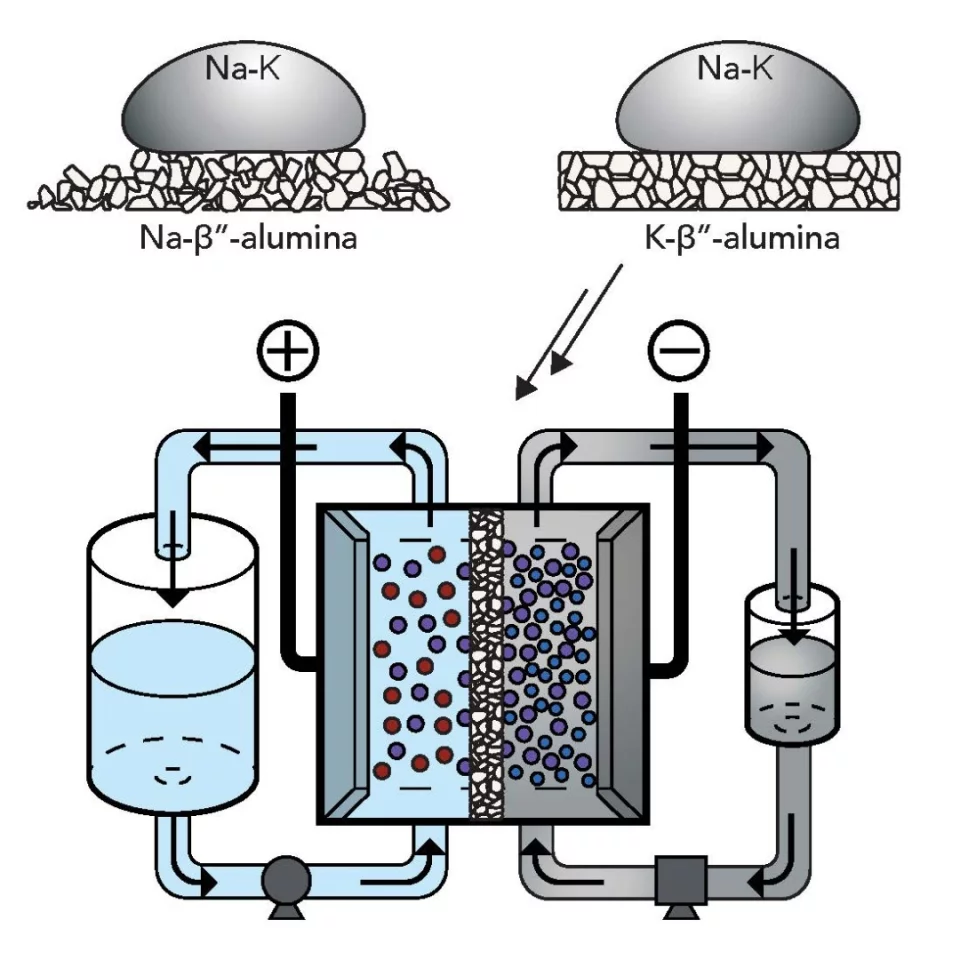
First and foremost, the fluid used as the negative side of the battery is an alloy of sodium and potassium. This mixture remains a liquid metal at room temperature, and theoretically packs at least 10 times the energy density of other fluids previously suggested for the role. On the positive side of the cell, the team tested four different water-based liquids.
The second new material is in the membrane used inside the cell. The team made a ceramic membrane out of potassium and aluminum oxide, which keeps the positive and negative fluids separate while still allowing current to flow between them.
The combination of the new anolyte and the new membrane, reportedly produces twice the maximum voltage of other flow batteries, which means a better overall energy density and lower production cost. The prototype the team developed also proved its stability over thousands of hours of operation.
"A new battery technology has so many different performance metrics to meet: cost, efficiency, size, lifetime, safety, etc.," says Antonio Baclig, co-author of the study. "We think this sort of technology has the possibility, with more work, to meet them all, which is why we are excited about it."
To further improve the battery design, the researchers say in future they could tweak the membrane thickness, or use a non-watery liquid for the positive side of the cell.
The research was published in the journal Joule.
Source: Stanford University










