Hydrogen is a clean fuel source, but current methods of producing it, often by converting natural gas, can undo any environmental benefit. Producing hydrogen out of sunlight and water doesn't create any CO2, and recent research has improved the efficiency and lowered the cost of devices that achieve this. Now, engineers from Columbia University are developing a "solar fuels rig" that floats on the ocean, captures energy through a solar cell and uses it to harvest hydrogen from the water beneath it.
The rig produces hydrogen through water electrolysis, a technique where H2 and O2 gases are separated out of water by passing an electric current through the liquid. Most of the time, these devices require a membrane to separate the two electrodes, but these membranes are fragile and require very pure water, which limits their practical applications.
The device developed at Columbia can split water into hydrogen and oxygen without needing a membrane. That means it can be deployed on seawater, which would normally degrade a membrane thanks to the impurities and micro-organisms that call it home.
"Being able to safely demonstrate a device that can perform electrolysis without a membrane brings us another step closer to making seawater electrolysis possible," says Jack Davis, the first author of a paper describing the device. "These solar fuels generators are essentially artificial photosynthesis systems, doing the same thing that plants do with photosynthesis, so our device may open up all kinds of opportunities to generate clean, renewable energy."
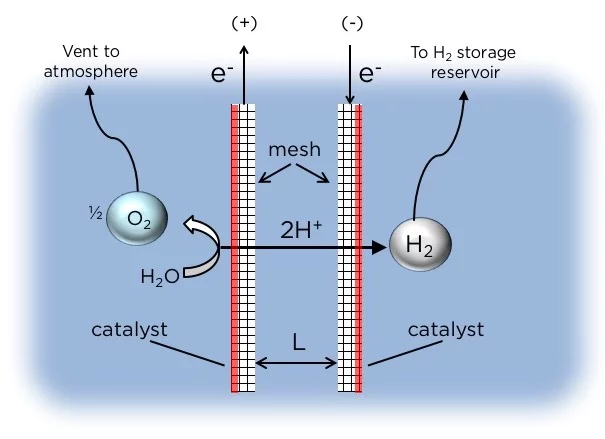
Instead of a membrane, the Columbia system uses two mesh flow-through electrodes that are designed to be asymmetric. Each one is coated with a catalyst only on the outer edge, and the bubbles of gas form on these surfaces. H2 bubbles form on one electrode and O2 on the other, and to harvest these gases, the device uses simple physics – namely, they wait for the bubbles to grow big enough that they float up to the surface. The O2 is allowed to bubble up to the surface and escape into the air, while the H2 bubbles float into a collection chamber.
This unique electrolysis mechanism is hooked up to a photovoltaic cell, which generates the required electric current with energy gathered from sunlight. The whole shebang can be mounted on a floating platform on the open sea.
The team is currently refining the design before testing it in real seawater, and eventually scaling up the system.
"We are especially excited about the potential of solar fuels technologies because of the tremendous amount of solar energy that is available," says Daniel Esposito, lead researcher on the project. "Our challenge is to find scalable and economical technologies that convert sunlight into a useful form of energy that can also be stored for times when the sun is not shining."
The research was published in the International Journal of Hydrogen Energy and the device is demonstrated in the video below.
Source: Columbia University






