Hydrogen sulfide famously stinks like rotten eggs, and contributes that eye-watering, low-hanging punch to the bouquet of the very worst farts. It's also toxic, corrosive, flammable and produced in large amounts as an expensive-to-treat by-product at petroleum refineries. Now, researchers have found an easy, profitable way to turn it into hydrogen.
The current method for dealing with this stuff – an unavoidable waste product when you're refining oil or natural gas – is to heat it up with air to between 800-1,100 °C (1,470-2,000 °F), then run it through a series of condensers, reheaters and catalytic reactors to convert it into sulfur and water in what's known as the Claus Process. The sulfur can be sold on, but the high temperatures involved make this an energy-hungry process.
Now, researchers at Rice University say they've found a way to pretty much eliminate that energy use and its associated emissions, while still recovering the sulfur and capturing useful hydrogen gas to boot.
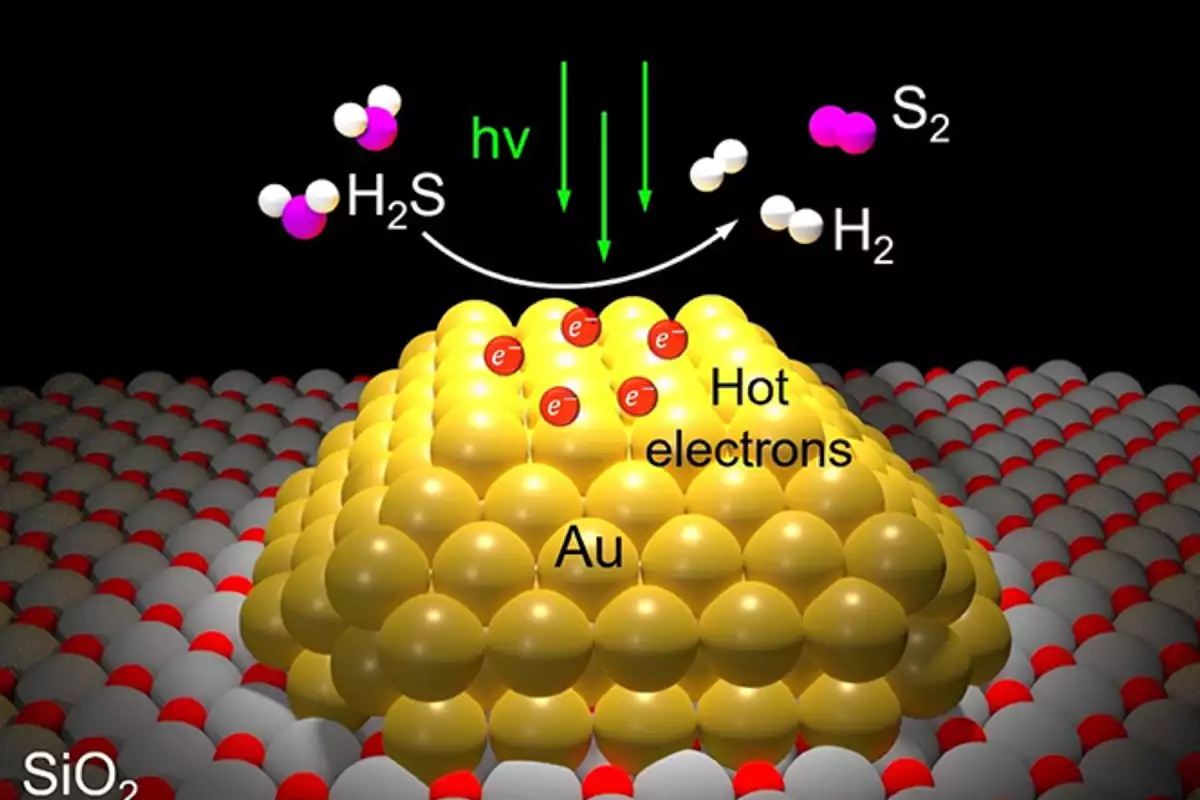
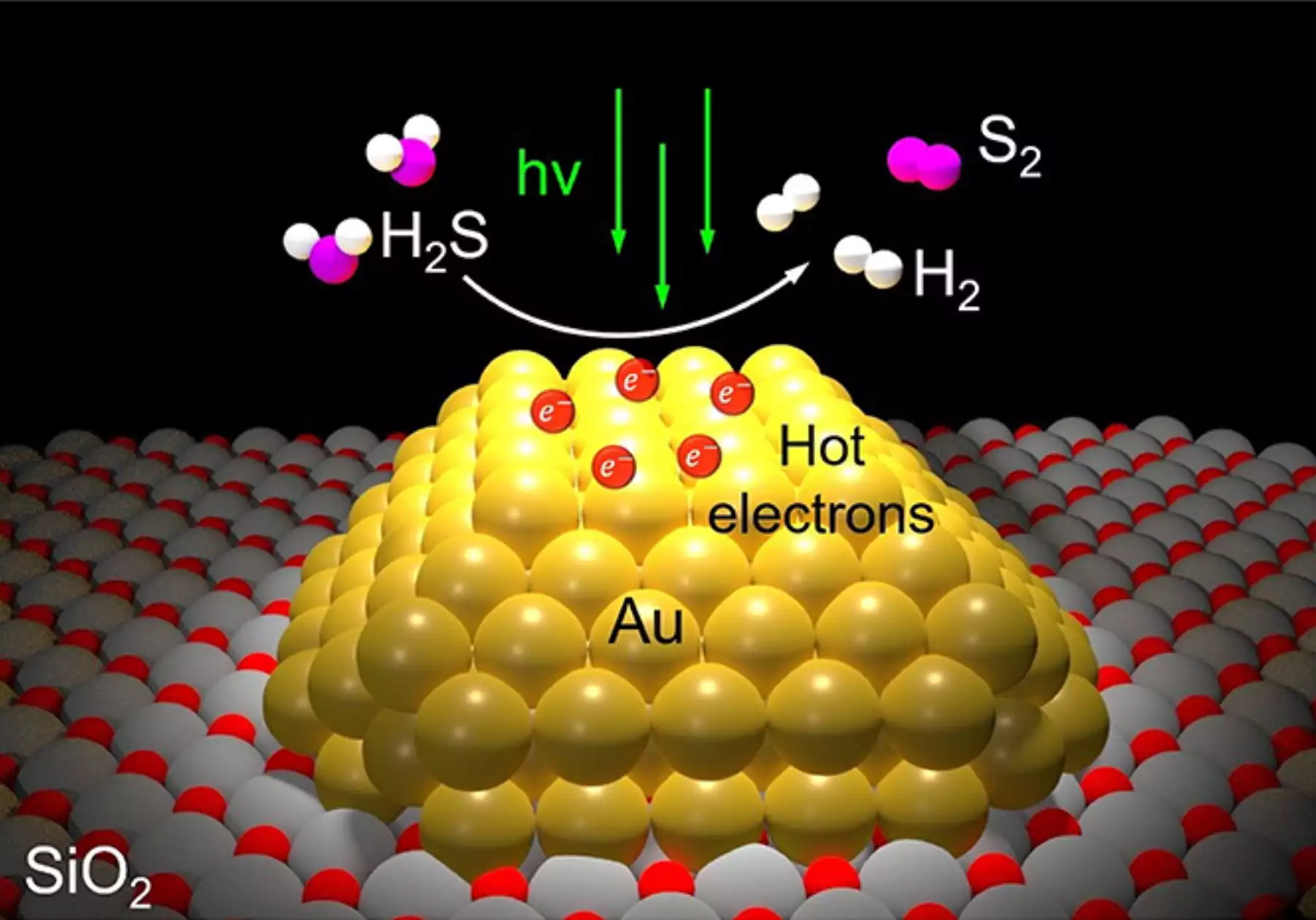
The new process uses a nano-engineered silicon dioxide powder catalyst, each grain speckled with nanoparticles of gold just billionths of a meter wide. These gold particles respond strongly to a specific wavelength of visible light, shooting out short-lived electrons known as "hot carriers," which carry enough energy to split H2S molecules efficiently into H2 and S for separate capture.
The process can be entirely powered by sunlight, and thus more or less free for a refinery in terms of operating costs where sunlight is available. But according to the research team, it works just fine with artificial light as well, and could end up being so cheap and efficient that you could roll it out with its own low-powered LED lighting and harvest hydrogen and sulfur while cleaning up sewer gas underground.
“Hydrogen sulfide emissions can result in hefty fines for industry, but remediation is also very expensive,” said Naomi Halas, lead author on the study and co-founder of Syzygy Plasmonics, which has licensed the new technology for commercialization. “The phrase ‘game-changer’ is overused, but in this case, it applies. Implementing plasmonic photocatalysis should be far less expensive than traditional remediation, and it has the added potential of transforming a costly burden into an increasingly valuable commodity.”
While this method should actually reduce carbon emissions due to the significant energy use it obviates in the refining process, it seems unlikely that the resulting hydrogen will meet the criteria for "green" classification, since at the end of the day it's coming from fossil fuel.
The study is available in the journal ACS Energy Letters.
Source: Rice University