A team of UK researchers has, for the first time in living human brains, demonstrated how the cognitive dysfunction seen in schizophrenia may be caused by the lack of a key protein known to facilitate communication between neurons in the brain.
“Our current treatments for schizophrenia only target one aspect of the disease – the psychotic symptoms – but the debilitating cognitive symptoms, such as loss of abilities to plan and remember, often cause much more long-term disability and there’s no treatment for them at the moment,” explains Oliver Howes, lead researcher on the new study. “Synaptic loss is thought to underlie these symptoms.”
For several decades researchers have hypothesized synaptic dysfunction as one of the key pathophysiological characteristics of schizophrenia. However, other than post-mortem brain examinations and animal experiments, there has been no way to investigate this hypothesis in living human subjects.
A genetic variation in the gene SV2A has previously been associated with increased schizophrenia risk. This gene plays a role in the production of a protein thought to aid in synaptic communication.
The research utilizes a newly developed radioactive PET tracer designed to specifically bind to SV2A proteins. This allows researchers to image the protein’s activity in a living brain for the first time.
Eighteen schizophrenic patients were scanned using the new imaging technology, and the results were compared with a control group of 18 subjects without schizophrenia. The results revealed the schizophrenic subjects showed significantly lower levels of SV2A in two key frontal brain regions: the frontal cortex and the anterior cingulate cortex.
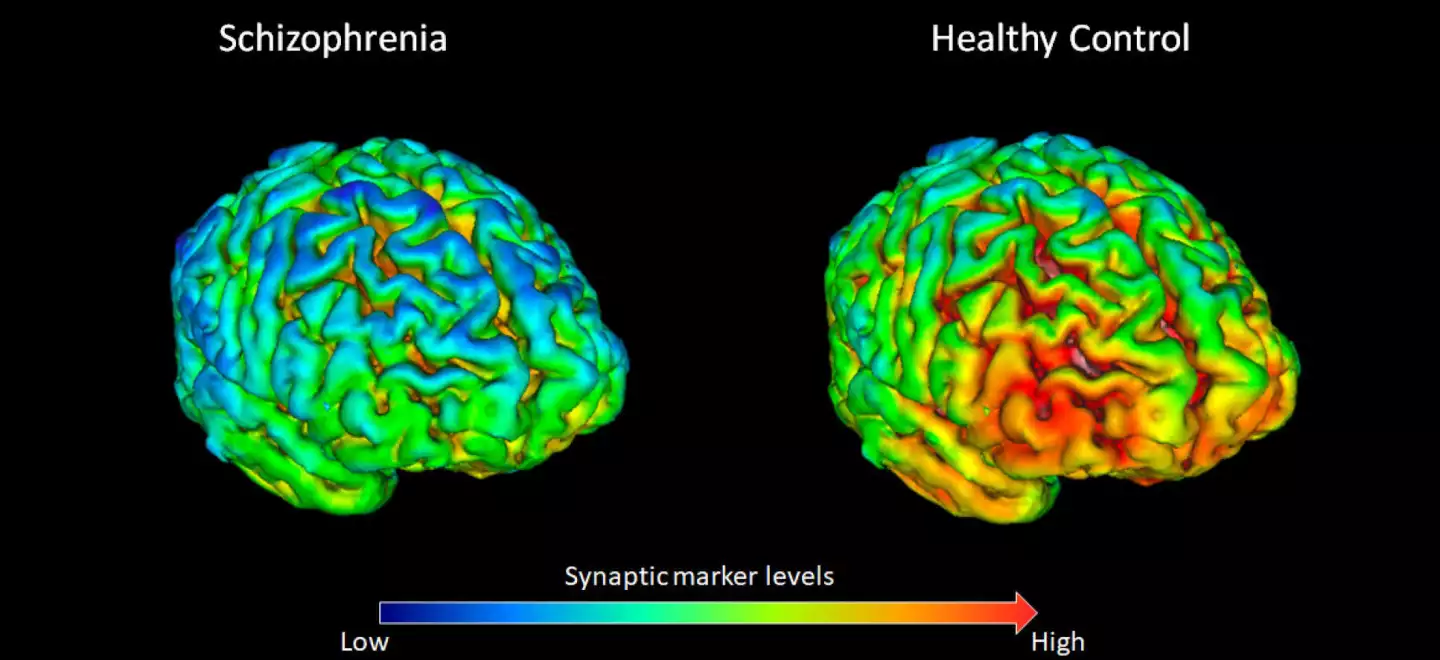
“Our lab at the MRC London Institute of Medical Sciences is one of the few places in the world with this new tracer," says Howes. “Which means we’ve been able for the first time to show there are lower levels of a synaptic protein in people with schizophrenia. This suggests that loss of synapses could underlie the development of schizophrenia.”
The study also importantly established that the two most common antipsychotic medications administered to schizophrenic patients were not the cause of this synaptic dysfunction. In subsequent rat studies the researchers affirmed both haloperidol and olanzapine did not affect SV2A protein levels.
“This is reassuring as it’s suggesting that our antipsychotic treatments aren’t leading to loss of brain connections,” notes Howes.
The discovery lays the track for future research pathways investigating novel schizophrenia treatments. Synaptic function in schizophrenic patients could be hypothetically restored by boosting SV2A activity, offering new therapeutic options to treat the cognitive symptoms seen in the disease.
The next step for the researchers will be using this new imaging technique to reveal what happens in human brains at the earliest stages of the disease.
“Next we hope to scan younger people in the very early stages to see how synaptic levels change during the development of the illness and whether these changes are established early on or develop over time,” says Howes.
The new study was published in the journal Nature Communications.
Sources: King’s College London, Imperial College London