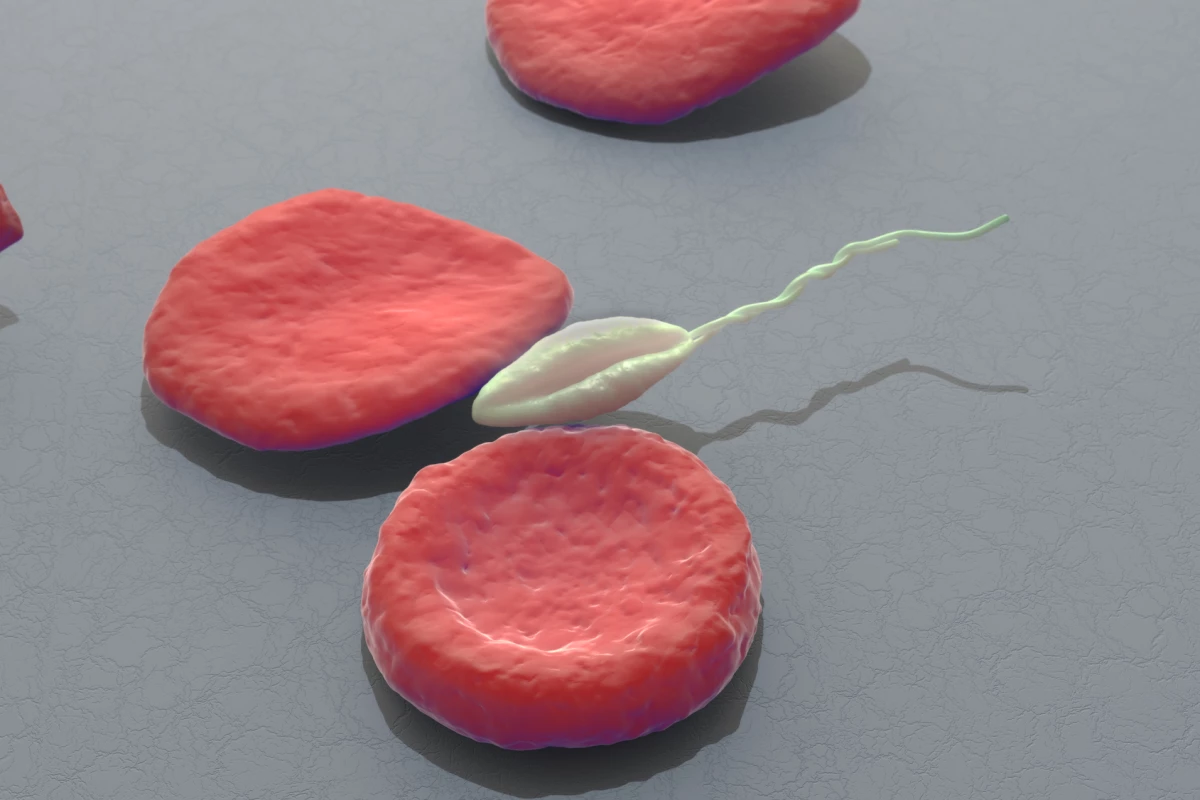
Every year around two million people suffer suffer from a skin disease called leishmaniasis, which is caused by the Leishmania major parasite. Humans have battled with this parasite for centuries, and some cultures have practiced a primitive form of inoculation to prevent infections.
Called leishmanization, this ancient remedy involves deliberately infecting a person with the live parasite. The process generates a minor skin infection which one's immune system can hopefully overcome, leaving the subject with life-long immunity to the parasite. However, this traditional treatment is rarely practiced today due to the tendency of patients to suffer from skin lesions than can linger for months.
In a wonderful example of blending an ancient treatment with cutting-edge 21st century technology, a team of scientists has gene edited the Leishmania parasite to limit its infectivity. This allows the parasite to be administered to patients in the form of a vaccine, creating long-term immunity without causing disease.
“Live vaccines like that are the best vaccines, but there’s a potential risk of causing serious disease in some people,” explains Abhay Satoskar, co-lead investigator on the new research, from Ohio State University. “We refined the concept using modern technology, making a parasite that does not cause clinical disease but allows for induction of immunity.”
Using CRISPR gene editing technology, the researchers removed two specific genes from the parasite to stop it producing a protein called centrin, which is responsible for supporting the parasite’s physical structure. When the parasite lacks centrin it can still enter host cells and replicate, but at a significantly slower rate that means infection does not lead to disease.
“So we’re essentially using leishmanization. CRISPR allowed us to do that,” Satoskar says. “The parasites are unable to proliferate, so they die. But they persist in the body for eight or nine months, which is long enough to generate acquired immunity.”
In their study, the researchers chronicle the extensive development and animal testing for this prospective novel vaccine. The study shows the CRISPR-edited parasites do not cause disease in animal models of sand flies, which spread the parasite through their bite, and mice. Plus, the mutant parasite effectively generated complete immunity in the majority of animal tests, including in mice with compromised immune systems.
“The multiple animal tests also made sure the genome didn’t revert back to normal,” adds Satoskar. “And we found that if a sand fly were to bite at the site of the vaccine and take mutated parasites into the wild, the parasites cannot survive. So it is environmentally safe.”
The researchers are now looking to begin Phase 1 human trials within the next two years. Satoskar also suggests the vaccine is likely to cost less than US$5 a dose, offering hope of an eventual cheap and easy roll out in those hardest hit tropical countries.
The study was published in the journal Nature Communications.
Source: Ohio State University