NASA's Perseverance Mars rover has notched up another first by extracting oxygen from the Martian atmosphere. On April 20, the toaster-sized Mars Oxygen In-Situ Resource Utilization Experiment (MOXIE) converted carbon dioxide into about 5.4 grams of oxygen – a process that may one day help astronauts live off the land on the Red Planet.
Current plans for the first crewed missions to Mars require a long stay on the surface until the planets are in the right position for the return journey. Unfortunately, this will require either an enormous amount of rocket fuel and supplies or some way has to be found to harvest resources on Mars.
It takes 7 tonnes of rocket fuel and 25 tonnes of oxygen to lift four astronauts from the surface of Mars into orbit, and each astronaut would need a tonne of oxygen each per year for breathing. That's far too much to bring from Earth because transporting the oxygen would require more fuel and oxygen, plus more fuel and oxygen to transport the additional fuel and oxygen, and more to transport that ...

On the plus side, though the Martian atmosphere is only 100th as dense as the Earth's, it's almost all carbon dioxide, which is made up of one carbon atom and two oxygen atoms. With the proper equipment and enough power available, it could be entirely practical to generate oxygen from Martian CO2.
To explore this possibility, Perseverance has been equipped with the MOXIE unit to demonstrate the technology that could support future missions. The device works by running the Martian atmosphere through a HEPA filter, then compressing and heating it to 1,470 °F (800 °C) with a scroll compressor. At this elevated temperature, a ceramic oxide splits the carbon dioxide into oxygen and carbon monoxide through a process called solid oxide electrolysis.
As to the unit itself, it's made of heat-tolerant materials and insulated with aerogels. The outer shell is coated in gold to reflect heat to protect the rest of the rover. Meanwhile, 3D-printed nickel parts help to transmit heat to and from the gases inside.
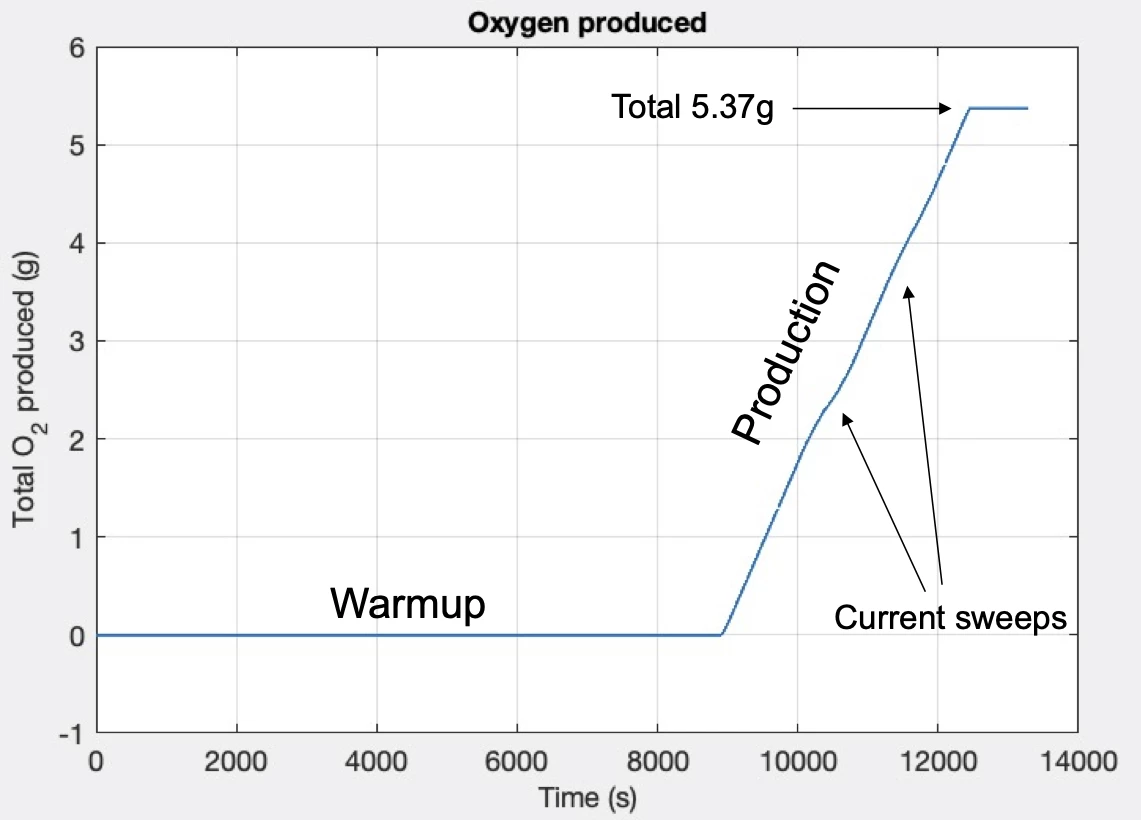
For the recent test, MOXIE went through a two-hour warmup before going into production mode, generating almost six grams of oxygen per hour in a series of sweeps. That's enough for an astronaut to breathe for 10 minutes. A future version of MOXIE weighing one tonne could crank out many tonnes of oxygen.
MOXIE is scheduled to carry out a series of tests in three phases over the next Martian year, or over two Earth years. The first phase will focus on testing the instrument itself. The second phase will work on producing oxygen under different atmospheric conditions, and the third phase will expand into new operating modes, like varying operating temperatures.
"MOXIE isn’t just the first instrument to produce oxygen on another world," says Trudy Kortes, director of technology demonstrations within STMD. It’s the first technology of its kind that will help future missions 'live off the land,' using elements of another world’s environment, also known as in-situ resource utilization.
"It’s taking regolith, the substance you find on the ground, and putting it through a processing plant, making it into a large structure, or taking carbon dioxide – the bulk of the atmosphere – and converting it into oxygen. This process allows us to convert these abundant materials into usable things: propellant, breathable air, or, combined with hydrogen, water."
Source: NASA






