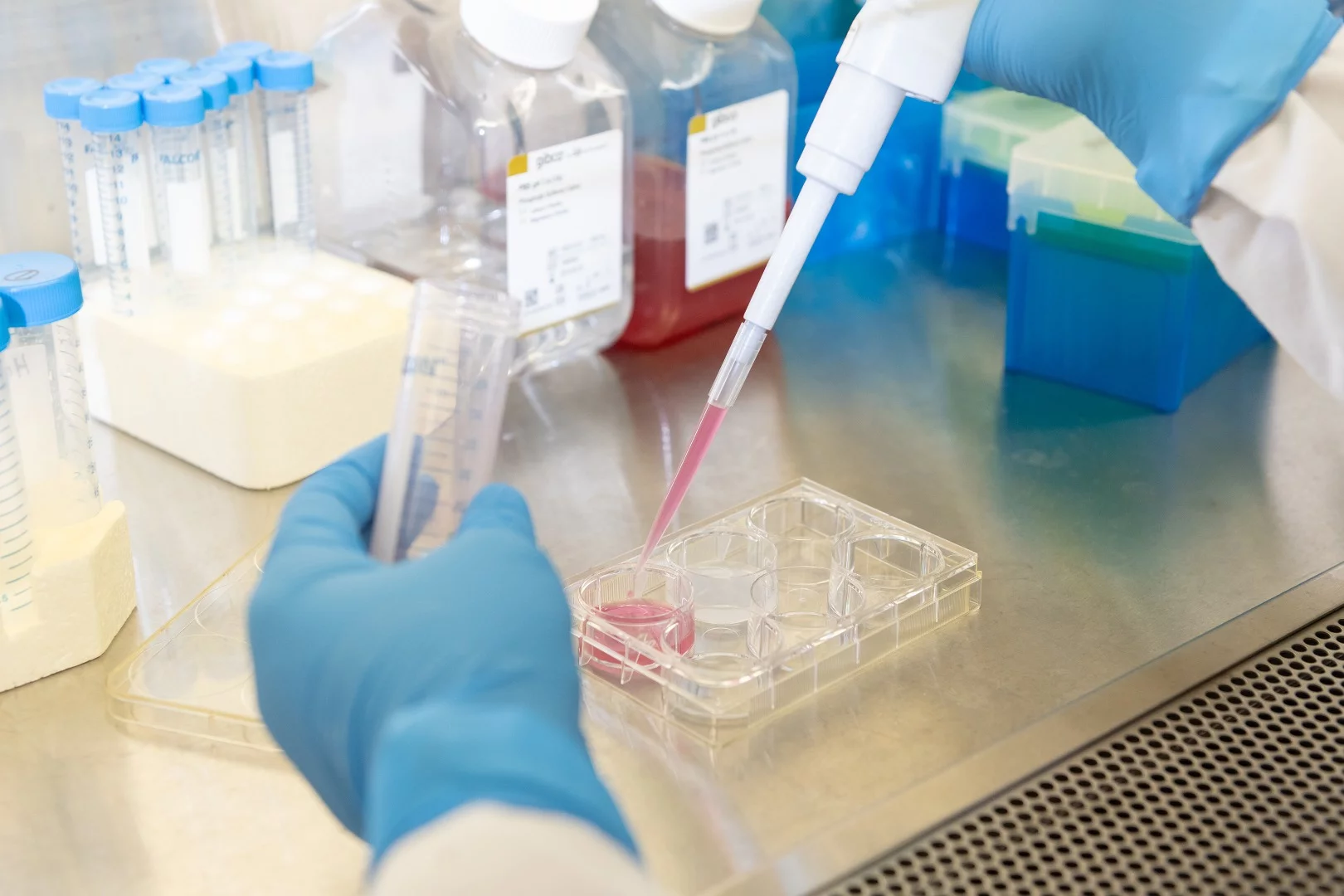
Cancer drugs are getting more effective at killing tumors, but there's one big problem that may be holding them back from wide use: many work like a shotgun blast and harm healthy cells as well. Researchers from Georgia Tech have now developed a new way to deliver drugs only to where they need to be, using what they describe as nanoscale "glass" bottles.
The design of the new drug delivery system is quite clever. The "bottles" are hollow spheres made of silica and measuring about 200 nanometers wide, with a hole in the surface. These spheres can be packed with basically any drug that's needed.
To keep the payload in place until it gets to the right spot, the drug is mixed with fatty acids and a near-infrared dye. When a doctor wants to release the drug, they simply shine an infrared laser on the relevant area. The dye in the spheres absorbs the heat from the laser and melts the fatty acids, releasing the medical payload through the hole. This prevents the drug from leaking into healthy cells where it can do harm.

"This new method could allow infusion therapies to target specific parts of the body and potentially negating certain side effects because the medicine is released only where there's an elevated temperature," says Younan Xia, corresponding author of the study. "The rest of the drug remains encapsulated by the solid fatty acids inside the bottles, which are biocompatible and biodegradable."
To make the capsules, the team starts with spheres of polystyrene with a gold nanoparticle embedded in it, which are then coated in a silica material. When the polystyrene and gold is removed, they're left with hollow silica spheres, and the size of the holes can be tweaked by using bigger or smaller gold particles.
Next, the bottles are loaded with the payload by soaking them in a mixture of the drug, fatty acids and dye. Any trapped air is removed and the excess material is then washed away with water.
The team tested the system by putting these nanocapsules in water, and tracking the levels of therapeutics in that water over time as they used an infrared laser on them. They found that when the laser wasn't used the drugs didn't escape into the water, but within a few minutes of heating, concentrations of the chemicals rose in the water.
If this kind of targeted approach sounds familiar, that's because this is just one of many techniques designed to release drugs only where and when they're needed. Others include a swallowable star-shaped capsule that releases multiple doses over a week, drugs bound together with DNA that degrades at different intervals, a Bluetooth-controlled implant, nanobubbles filled with chemotherapy drugs that are burst with X-rays, and even bacteria that carry drugs and release them on demand through ultrasound pulses.
The new capsules sound like a promising addition to this list, but of course they'll first need to be tested in vivo and eventually on humans.
"This controlled release system enables us to deal with the adverse impacts associated with most chemotherapeutics by only releasing the drug at a dosage above the toxic level inside the diseased site," says Jichuan Qiu, an author of the study.
The research was published in the journal Angewandte Chemie International Edition.
Source: Georgia Tech