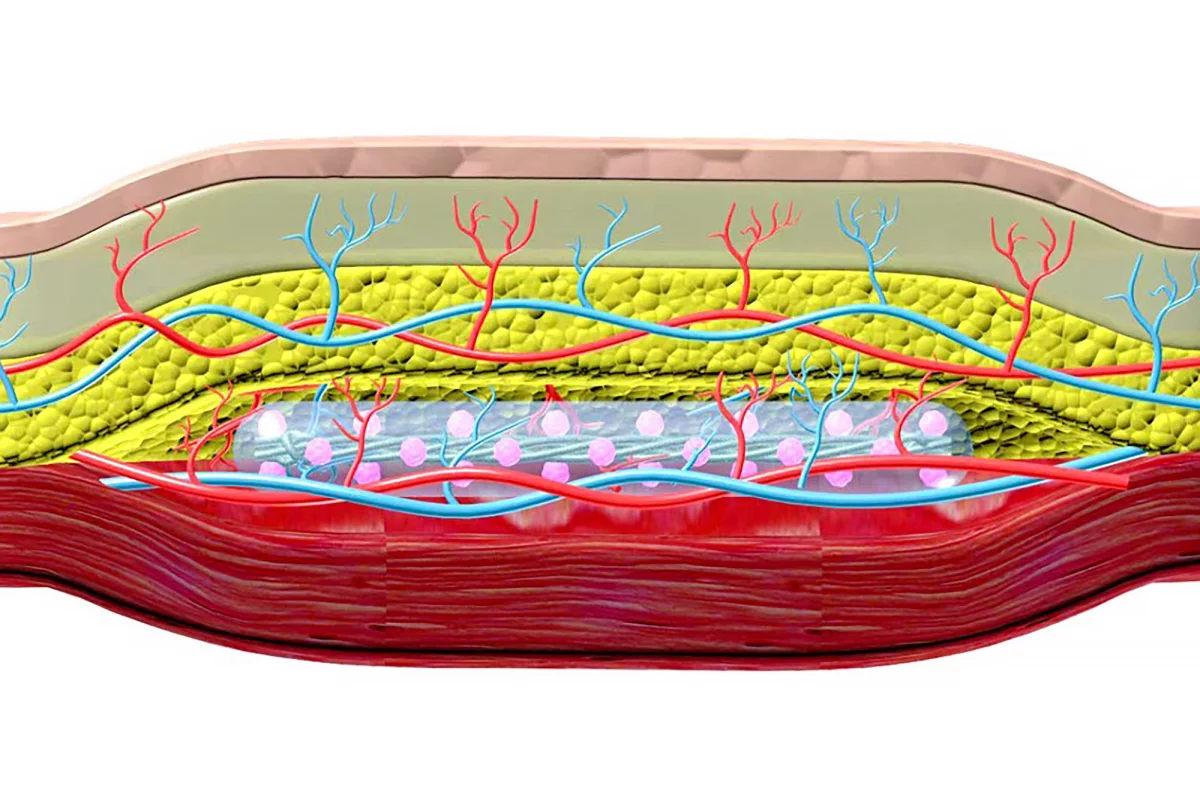
Researchers have developed a technique to implant a thread-like device containing insulin-secreting pancreatic cells in a pocket under the skin. The device reversed type 1 diabetes in mice and didn’t require the administration of anti-rejection drugs. The device could one day provide an alternative to insulin injections.
In type 1 diabetes, the body’s immune system attacks and destroys the insulin-producing cells of the pancreas, called islet cells, preventing the secretion of insulin and necessitating the lifelong administration of insulin injections or the use of an insulin pump.
Researchers from Cornell University and the University of Alberta (U of A) collaborated to create an under-the-skin implant that secretes insulin while avoiding the immune reactions implanted devices can produce.
“Over the years, I receive a lot of emails and requests from parents and patients saying, ‘Hey, my baby was diagnosed with type 1 [diabetes], can you help us?’” said Minglin Ma, one of the study’s corresponding authors. “It’s a very bad disease, and a lot of children have it. So, we are really serious about pushing this into something clinically applicable, something that’s impactful.”
In 2017, Ma, from Cornell’s College of Agriculture and Life Sciences (CALS), developed the Thread-Reinforced Alginate Fiber For Islets enCapsulation (TRAFFIC), a removable nylon thread implant containing hundreds of thousands of islet cells protected by a thin alginate hydrogel coating and inserted into the abdominal cavity. A more robust version of the implant was created in 2021 that effectively controlled blood sugar in mice for up to six months.
Ma’s implant attracted the attention of U of A diabetes researcher James Shapiro, who’d created a method for inserting islet cells in channels just under the skin and then applying immunosuppression to protect them.
“I was intrigued by the virtue of Ma’s approach as it avoided the need for immunosuppression, and I wondered if we might combine our two innovative strategies to improve cell survival,” Shapiro, the study's other corresponding author, said. “And indeed, it worked! By combining the two it really did improve the skin site for engrafting cells without the need for anti-rejection drugs.”
The resulting collaboration produced SHEATH, the Subcutaneous Host-Enabled Alginate THread.
SHEATH implantation is a two-step process. First, a medical-grade nylon catheter is inserted under the skin, where it remains for four to six weeks. The catheter triggers a controlled foreign-body inflammatory response that causes a dense network of blood vessels to form around it. When the catheters are removed, the alginate-based islet cell-seeded device is inserted into the pocket – or channel – that’s been created, with the surrounding vessels providing the islet cells with the required oxygen and nutrients.
“That channel is a perfect fit for our device,” Ma said. “Shapiro used the analogy that this is like a hand in a glove. And putting something under your skin is much easier, much less invasive than in the abdomen. It can be done as an outpatient procedure, so you don’t have to stay in hospital. It can be done under local anesthesia.”
Implanting the SHEATH system in diabetic mice led to a reversal of the condition without the need for immunosuppression. Experiments showed a robust capacity for long-term diabetes reversal, with some mice achieving correction of high blood glucose for over 190 days. Moreover, the system allowed for the removal and replacement of a failing implant, indicated by rising blood glucose levels. After being replaced with a fresh implant, blood glucose levels returned to normal.
Confirming the system’s scalability, the researchers successfully developed procedures to implement the SHEATH approach in minipigs, including insertion, removal, and replacement of the implant.
The researchers acknowledge that, despite the SHEATH system’s promising features, additional challenges regarding its clinical utility need to be overcome. Specifically, determining an acceptable catheter length and identifying anatomically appropriate sites for placement.
“The challenge is, it’s very difficult to keep these islets functional for a long time inside of the body where you have a device because the device blocks the blood vessels, but the native islet cells in the body are known to be in direct contact with vessels that provide nutrients and oxygen,” said Ma. “The device is designed in a way that we can maximize the mass exchange of nutrients and oxygen, but we may need to provide additional means to support the cells for a long-term function in large-animal models and eventually patients.”
These ‘additional means’ may include incorporating a continuous oxygen supply into the device. Ma has formed a new Cornell spin-off, Persista Bio, to develop a separate device to supply additional oxygen to the cells.
Despite these challenges, the researchers are hopeful that future versions of the implant device will be able to last for two to five years before needing to be replaced.
The study was published in the journal Nature Biomedical Engineering.
Source: Cornell University, University of Alberta