It’s a cruel reality that the heart has such a limited capacity to heal itself, meaning that people who survive heart attacks often go on to have more. Now, researchers at University College London (UCL) have grown heart cells from stem cells, embedded them onto microspheres and injected them into damaged hearts to help repair them.
After a heart attack, the vital organ patches up the damage with scar tissue, which keeps its structure intact but at the crucial expense of rhythm. Because this tissue doesn’t beat, eventually the stress can lead to heart failure.
The problem seems to be that heart stem cells don’t exist, so heart muscle cannot be replenished. That complicates stem cell therapies, which have proven promising in regenerative medicine in other parts of the body. Some success has been found using more general stem cells, but frustratingly they seem to have a hard time staying in the right place in the heart long enough to get to work.
And that’s the issue that the new study set out to address. Other teams have attempted to keep stem cells in place by embedding them in hydrogels or combining heart muscle and supportive cells, but this time the UCL researchers used biodegradable microspheres.
Each measuring just a quarter of a millimeter wide, these microspheres have a very porous structure, giving cells plenty of surface area to attach to. Heart cells are then grown from stem cells on these microspheres, where they’re able to form connections and develop into new heart tissue that can beat. The spheres are small enough that they could be injected into heart muscle.
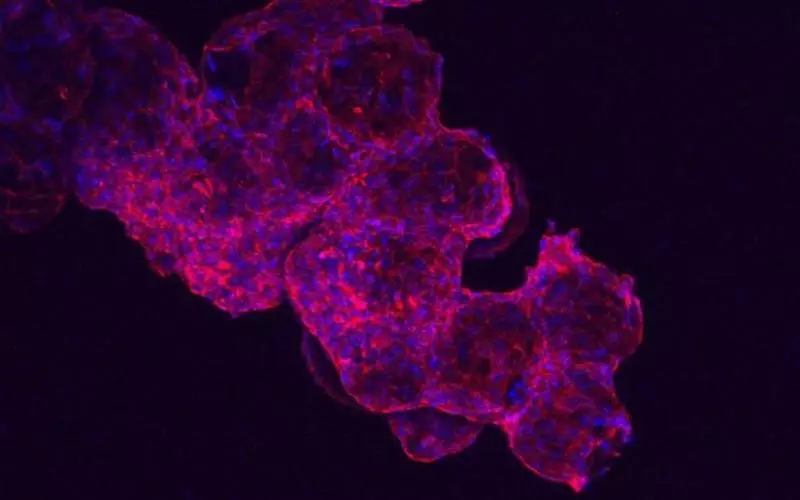
In lab tests, the new cells were able to beat for up to 40 days in a dish, and when injected into rat hearts they stayed in place for up to six days. Obviously that still leaves a lot of room for improvement, but it’s a promising start.
“To truly mend broken hearts, it is important that stem cells are delivered in a way that allows them to survive within their new environment and turn into heart muscle cells,” says Daniel Stuckey, lead researcher on the study. “Our technology provides a new way of ensuring that the cells injected into the heart are working as they should. We hope this research will give us the information essential for making stem cell therapy a realistic treatment for people with heart disease.”
Other recent research has tackled the problem of repairing damaged hearts by using things like cardiac progenitor cells, placental stem cells, stem cell “messengers” called exosomes, or healing cells in the fluid around the heart, while some have reprogrammed structural cells into ones that beat.
The new research was presented at the British Cardiovascular Society Conference.
Source: UCL




